Now I want to discuss a few reactions that add hydrogens to triple bonds. And these are going to be called, in general, hydrogenation reactions. The major difference between them is going to be how many hydrogens they add and in what stereochemistry. So let's go ahead and check them out. Basically, there are 3 major types of hydrogenation that we look at and these are going to be reactions that turn triple bonds into either double bonds or single bonds. So basically we're taking one type of hydrocarbon and making it a more saturated version of that hydrocarbon. Remember that the definition of saturation has to do with how many hydrogens are on that molecule. Okay? So in all of these reactions, what we're doing is we're increasing the saturation of the molecule, meaning that we're adding hydrogens to it.
Hydrogenation of Alkynes - Online Tutor, Practice Problems & Exam Prep
Hydrogenation reactions are a subtype of reduction reactions that add hydrogens to double and triple bonds. These differ in the types of products made and the stereochemistry of the mechanism.
The definition of hydrogenation.
Video transcript
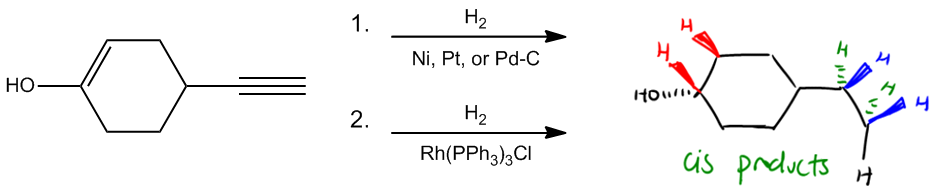
Using Catalytic hydrogenation or Wilkinson’s Catalyst to turn alkynes to alkanes.
Video transcript
So let's look at the first and probably simplest form of hydrogenation, which we call full saturation. What that means is that I'm taking a double bond or a triple bond and I'm fully saturating it with hydrogens. Alright? We can do this using two different reagents. We can achieve this with either catalytic hydrogenation, which involves the reagents shown here in the first arrow. That would be basically H 2 over a plate of platinum, palladium, or nickel catalyst. Okay? Or there's another reaction we can use called Wilkinson's catalyst. And Wilkinson's catalyst is used for the same purpose: to take double bonds and triple bonds and fully saturate them to alkanes. Alright? As you can see, the reagents for Wilkinson's catalyst are a bit confusing. It's got the H 2 because we're adding hydrogen of course, but the catalyst is way different. In this case, we're going to use rhodium with three triphenylphosphines and then a chlorine. It looks messy. Okay? I'm not going to draw the whole thing for you as long as you can recognize that that's Wilkinson's, that's fine. Okay?
The only thing about full saturation is that this is a bit of a misnomer because remember that rings also count towards unsaturation. A ring is something that's missing two hydrogens because two ends are attached to each other. And actually, these reactions do nothing to rings. So what's going to happen is that all the double bonds and triple bonds will be gone, but the rings will stay intact. Alright?
One more thing before I draw these products. Notice that the stereochemistry mentions syn addition. Okay? Syn addition is another way to say that we're going to get cis products. So, what that means is that I would expect the hydrogens that we're adding to add from the same side of the double bond or the same side of the triple bond. So let's go ahead and draw our products. Basically, I would get the same exact sigma framework. When I say sigma framework, I'm just saying all the sigma bonds are the same. But now I would get a single bond over here, so that triple bond just turned into an alkane. And I would get that this alcohol is still here. And then I would finally get that the double bond is also gone, meaning that there are no pi bonds left. Okay? This is really good at getting rid of pi bonds. That's pretty much it.
Here I did include stereochemistry because there are no chiral centers, so I don't really have to. But if you're wondering where these hydrogens came from, especially maybe on the double bond, they could have both come from the same side. So that means let's say that one of the hydrogens came from here and one came from here. Notice that they came from the same side of the double bond, then that means that my alcohol will be forced downward. Alternatively, I could have also had the hydrogens add from the bottom, which means that the OH would have gone up as well. The same thing with the triple bond, except that for the triple bond, I added four hydrogens. I didn't just add two. So I basically added, let's say, two from the front, H 2 , and I also added two from the back, H 2 . So what I wound up getting was instead of, and then also, now this carbon here makes sense. It has CH2. But this one looks like it doesn't make sense because it's also CH2. But remember, there was already one H to begin with on that triple bond. So that last H is still there and it's right next to my head. So does that make sense? Basically, I added the two blue hydrogens, then I added two green hydrogens, and then this black H was just always there. Alright? So basically, you can think that full saturation, with either one of these two catalytic methods or Wilkinson's, is going to completely get rid of all pi bonds, all double bonds, and all triple bonds. Easy. Right? Cool.
Catalytic Hydrogenation or Wilkinson’s Catalyst: Alkane products
Stereochemistry: Syn Addition
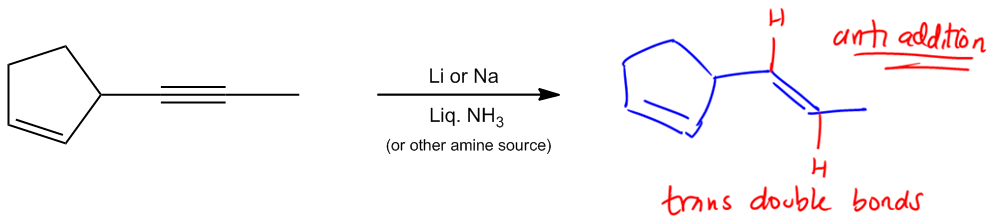
Using Dissolving Metal Reduction or Lindlar’s Catalyst to turn alkynes to alkenes.
Video transcript
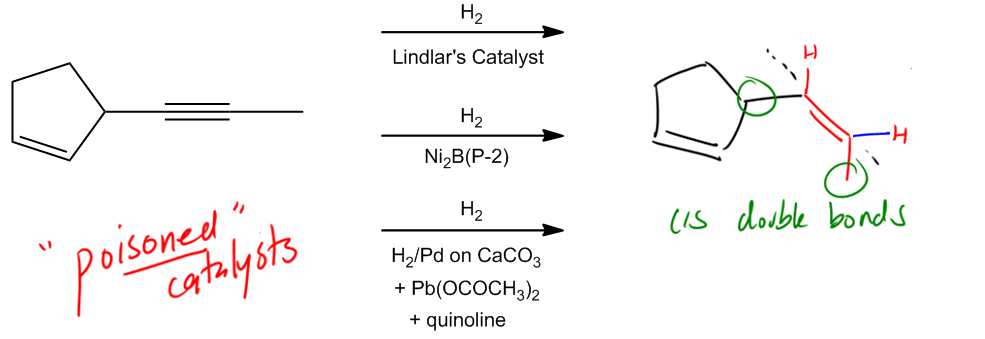
Move on to the next reaction. And actually, these next two are both considered partial saturation. So what that means is that for these next two, I'm not saturating all the way to an alkane. In fact, what I'm doing is I'm taking a triple bond and I'm only reacting it to a double bond. So I am adding two hydrogens, but I'm stopping there. Okay? And the first of these, really the biggest difference between them, other than the reagents, which are totally different, are the stereochemistry. Notice that my first one is anti addition. Notice that my second one is syn addition. That's going to have a difference in what my products look like. So let's look at this first one. The first one is called dissolving metal reduction. Okay? Dissolving metal reduction is just another name for these reagents. And what it is is that we have some kind of metal, lithium or sodium. Your professor can use either one in some kind of liquid amine source. Many times that amine is going to be NH3, but there's a lot of other things it could be. It could be NH2CH3 or it could be just any kind of usually primary or secondary amine. Alright? Cool. So we've got that part down. What does it do? Well, we learned I just taught you guys that partial saturation means that it's only working on triple bonds. So notice that I have the same or a very similar molecule here and what I'm wondering is how should I draw this? I have the same sigma framework, but for example, should I draw the double bond, or should I leave it there? I should leave it there because it turns out that since this is partial saturation, it has no effect on alkenes. No effect on alkenes. Alright? It's only going to work on triple bonds. So now I've got this triple bond. Is it going to work on this one? Absolutely. Well, what does anti addition mean? Anti addition, if you remember, that just means I have trans products. Okay? So what that means is that I would expect that my hydrogens are going to add from different sides of this double bond. What that means is that I should actually draw the double bond in a transposition. Why? Because the two hydrogens that I added must have added on different sides. Okay? Of different sides of the double bond, which means that they were anti addition. Is that making sense? So what that means is that dissolving metal reduction is important because this is a way that we can turn triple bonds into trans double bonds. That's going to be your product. Trans double bonds. Pretty cool, right? So that's something you have to keep in mind because, for synthesis later, when we start trying to make molecules, that's going to be the only way that we know how to turn a Sherbob wand and make it trans. Cool? So now let's go to this next one, which I'm sure you guys can already kind of anticipate what's going to happen. Notice that this one is called Lindlar's catalyst. And Lindlar's catalyst has a lot of different reagents that you could use. Unfortunately, different textbooks, different professors have their own way of writing it. Some professors are going to be really easy. They're going to be like, You know what? F all these reagents. We're just going to write Lindlar's. Okay? They're going to put H2 in Lindlar's, and you're going to know that that's Lindlar's. But some professors are going to use the actual reagents that are in the textbook, so some textbooks have this as the P2 catalyst. Okay? So the P2 catalyst is one form of Lindlar's. Another form is actually with, lead acetate, and quinoline. These are reagents that you just have to recognize that they are Lindlar's. You don't need to know exactly what they look like or draw them or even know the mechanism. But what you do need to know is to recognize that these are Lindlar's. These are also, just so you guys know, another term that you may hear is that these are poisoned catalysts. Okay? Now what could oops, Catalysts. Wow. Okay. Sorry, guys. Spelling isn't my forte. So poison catalyst. What does poisoned mean? What it means is that these catalysts have been created in such a way that they're not going to reduce all the way, or they're not going to hydrogenate all the way. Instead, they're going to stop at a given point.
Dissolving Metal Reduction: Alkene products
Stereochemistry: Anti Addition
Lindlar’s Catalyst: Alkene products
Stereochemistry: Syn Addition
Predict the product of the multistep synthesis





Do you want more practice?
More setsYour Organic Chemistry tutors
- Answer Problem 39 , parts a–h, using 2-butyne as the starting material instead of propyne. h. H2/Lindlar cata...
- Answer Problem 39 , parts a–h, using 2-butyne as the starting material instead of propyne. g. excess H2, Pd/C
- What are products of the following reactions? b.
- What are products of the following reactions? a.
- What stereoisomers are obtained from the following reactions? b. CH3CH2C☰CCH2CH3→1.H2/Lindlar catalyst 2. D2...
- What stereoisomers are obtained from the following reactions? a. CH3CH2C☰CCH2CH3→1.Na, NH3(liq), −78°C 2. D2...
- (•••) LOOKING AHEAD CHAPTER 10 The following reaction was recently reported to have been performed electrochem...
- Show how you would convert a. oct-3-yne to cis-oct-3-ene. b. pent-2-yne to trans-pent-2-ene.
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- Predict the product of the following alkyne reductions. (a)
- Predict the product of the following alkyne reductions. (b)
- Predict the product of the following alkyne reductions. (c)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- When sodium generates electrons in the presence of ammonia, these electrons persist in solution, giving the bl...
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Hydrogenation of which alkynes would produce the following cis-alkenes? (c)
- The following deprotonation step occurs during the trans reduction of an alkyne. Calculate K_eq for this react...
- Sodium amide, the base we use to deprotonate terminal alkynes, is synthesized by reducing ammonia via a mechan...
- Reduction of an alkyne using the Lindlar catalyst, a reaction presented in Section 10.6.2, produces only the c...
- Identify the product when each of the following reactions is performed on the triglyceride of linoleic acid (l...
- Hydrogenation of which alkynes would produce the following cis-alkenes?(b) <IMAGE>
- Long, polarized bonds are also reducible in the same way that the C―C π bond of an alkyne is. Show a mechanism...
- What is the major product of the reaction of 1 mol of propyne with each of the following reagents?g. excess H2...
- Describe the alkyne you should start with and the reagents you should use if you want to synthesizec. trans-2-...
- What reagents would you use for the following syntheses?c. hexane from 3-hexyne
- How can the following compounds be prepared using ethyne as the starting material?e. <IMAGE>
- Show how each of the following compounds can be prepared using the given starting material, any needed inorgan...
- How can the following compounds be prepared using ethyne as the starting material?f. <IMAGE>
- What are products of the following reactions?c. <IMAGE>d. <IMAGE>
- Show how you would convertc. cis-cyclodecene to trans-cyclodecene.
- What reaction would acetylene likely undergo if it were kept at 1500 °C for too long?
- Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or termi...
- Predict the product of the following reactions. (b)
- Describe the alkyne you should start with and the reagents you should use if you want to synthesize b. cis-2-...
- Describe the alkyne you should start with and the reagents you should use if you want to synthesize a. pentan...
- We have studied a number of pericyclic reactions previously. Draw the mechanism of the steps shown. The sectio...
- What alkene would you start with if you wanted to synthesize b. ethylcyclopentane?
- a. How many alkenes could you treat with H2, Pd/C to prepare methylcyclopentane? b. Which of the alkenes is t...
- What reagents would you use for the following syntheses? a. (Z)-3-hexene from 3-hexyne
- What reagents would you use for the following syntheses? b. (E)-3-hexene from 3-hexyne
- Predict the product of the following hydrogenation reactions. (b)
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (f) < of re...
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (i) < of re...
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (g) < of re...

