Now I want to discuss a few reactions that add hydrogens to triple bonds. And these are going to be called, in general, hydrogenation reactions. The major difference between them is going to be how many hydrogens they add and in what stereochemistry. So let's go ahead and check them out. Basically, there are 3 major types of hydrogenation that we look at and these are going to be reactions that turn triple bonds into either double bonds or single bonds. So basically we're taking one type of hydrocarbon and making it a more saturated version of that hydrocarbon. Remember that the definition of saturation has to do with how many hydrogens are on that molecule. Okay? So in all of these reactions, what we're doing is we're increasing the saturation of the molecule, meaning that we're adding hydrogens to it.
- 1. A Review of General Chemistry5h 5m
- Summary23m
- Intro to Organic Chemistry5m
- Atomic Structure16m
- Wave Function9m
- Molecular Orbitals17m
- Sigma and Pi Bonds9m
- Octet Rule12m
- Bonding Preferences12m
- Formal Charges6m
- Skeletal Structure14m
- Lewis Structure20m
- Condensed Structural Formula15m
- Degrees of Unsaturation15m
- Constitutional Isomers14m
- Resonance Structures46m
- Hybridization23m
- Molecular Geometry16m
- Electronegativity22m
- 2. Molecular Representations1h 14m
- 3. Acids and Bases2h 46m
- 4. Alkanes and Cycloalkanes4h 19m
- IUPAC Naming29m
- Alkyl Groups13m
- Naming Cycloalkanes10m
- Naming Bicyclic Compounds10m
- Naming Alkyl Halides7m
- Naming Alkenes3m
- Naming Alcohols8m
- Naming Amines15m
- Cis vs Trans21m
- Conformational Isomers13m
- Newman Projections14m
- Drawing Newman Projections16m
- Barrier To Rotation7m
- Ring Strain8m
- Axial vs Equatorial7m
- Cis vs Trans Conformations4m
- Equatorial Preference14m
- Chair Flip9m
- Calculating Energy Difference Between Chair Conformations17m
- A-Values17m
- Decalin7m
- 5. Chirality3h 39m
- Constitutional Isomers vs. Stereoisomers9m
- Chirality12m
- Test 1:Plane of Symmetry7m
- Test 2:Stereocenter Test17m
- R and S Configuration43m
- Enantiomers vs. Diastereomers13m
- Atropisomers9m
- Meso Compound12m
- Test 3:Disubstituted Cycloalkanes13m
- What is the Relationship Between Isomers?16m
- Fischer Projection10m
- R and S of Fischer Projections7m
- Optical Activity5m
- Enantiomeric Excess20m
- Calculations with Enantiomeric Percentages11m
- Non-Carbon Chiral Centers8m
- 6. Thermodynamics and Kinetics1h 22m
- 7. Substitution Reactions1h 48m
- 8. Elimination Reactions2h 30m
- 9. Alkenes and Alkynes2h 9m
- 10. Addition Reactions3h 18m
- Addition Reaction6m
- Markovnikov5m
- Hydrohalogenation6m
- Acid-Catalyzed Hydration17m
- Oxymercuration15m
- Hydroboration26m
- Hydrogenation6m
- Halogenation6m
- Halohydrin12m
- Carbene12m
- Epoxidation8m
- Epoxide Reactions9m
- Dihydroxylation8m
- Ozonolysis7m
- Ozonolysis Full Mechanism24m
- Oxidative Cleavage3m
- Alkyne Oxidative Cleavage6m
- Alkyne Hydrohalogenation3m
- Alkyne Halogenation2m
- Alkyne Hydration6m
- Alkyne Hydroboration2m
- 11. Radical Reactions1h 58m
- 12. Alcohols, Ethers, Epoxides and Thiols2h 42m
- Alcohol Nomenclature4m
- Naming Ethers6m
- Naming Epoxides18m
- Naming Thiols11m
- Alcohol Synthesis7m
- Leaving Group Conversions - Using HX11m
- Leaving Group Conversions - SOCl2 and PBr313m
- Leaving Group Conversions - Sulfonyl Chlorides7m
- Leaving Group Conversions Summary4m
- Williamson Ether Synthesis3m
- Making Ethers - Alkoxymercuration4m
- Making Ethers - Alcohol Condensation4m
- Making Ethers - Acid-Catalyzed Alkoxylation4m
- Making Ethers - Cumulative Practice10m
- Ether Cleavage8m
- Alcohol Protecting Groups3m
- t-Butyl Ether Protecting Groups5m
- Silyl Ether Protecting Groups10m
- Sharpless Epoxidation9m
- Thiol Reactions6m
- Sulfide Oxidation4m
- 13. Alcohols and Carbonyl Compounds2h 17m
- 14. Synthetic Techniques1h 26m
- 15. Analytical Techniques:IR, NMR, Mass Spect7h 3m
- Purpose of Analytical Techniques5m
- Infrared Spectroscopy16m
- Infrared Spectroscopy Table31m
- IR Spect:Drawing Spectra40m
- IR Spect:Extra Practice26m
- NMR Spectroscopy10m
- 1H NMR:Number of Signals26m
- 1H NMR:Q-Test26m
- 1H NMR:E/Z Diastereoisomerism8m
- H NMR Table24m
- 1H NMR:Spin-Splitting (N + 1) Rule22m
- 1H NMR:Spin-Splitting Simple Tree Diagrams11m
- 1H NMR:Spin-Splitting Complex Tree Diagrams12m
- 1H NMR:Spin-Splitting Patterns8m
- NMR Integration18m
- NMR Practice14m
- Carbon NMR4m
- Structure Determination without Mass Spect47m
- Mass Spectrometry12m
- Mass Spect:Fragmentation28m
- Mass Spect:Isotopes27m
- 16. Conjugated Systems6h 13m
- Conjugation Chemistry13m
- Stability of Conjugated Intermediates4m
- Allylic Halogenation12m
- Reactions at the Allylic Position39m
- Conjugated Hydrohalogenation (1,2 vs 1,4 addition)26m
- Diels-Alder Reaction9m
- Diels-Alder Forming Bridged Products11m
- Diels-Alder Retrosynthesis8m
- Molecular Orbital Theory9m
- Drawing Atomic Orbitals6m
- Drawing Molecular Orbitals17m
- HOMO LUMO4m
- Orbital Diagram:3-atoms- Allylic Ions13m
- Orbital Diagram:4-atoms- 1,3-butadiene11m
- Orbital Diagram:5-atoms- Allylic Ions10m
- Orbital Diagram:6-atoms- 1,3,5-hexatriene13m
- Orbital Diagram:Excited States4m
- Pericyclic Reaction10m
- Thermal Cycloaddition Reactions26m
- Photochemical Cycloaddition Reactions26m
- Thermal Electrocyclic Reactions14m
- Photochemical Electrocyclic Reactions10m
- Cumulative Electrocyclic Problems25m
- Sigmatropic Rearrangement17m
- Cope Rearrangement9m
- Claisen Rearrangement15m
- 17. Ultraviolet Spectroscopy51m
- 18. Aromaticity2h 34m
- 19. Reactions of Aromatics: EAS and Beyond5h 1m
- Electrophilic Aromatic Substitution9m
- Benzene Reactions11m
- EAS:Halogenation Mechanism6m
- EAS:Nitration Mechanism9m
- EAS:Friedel-Crafts Alkylation Mechanism6m
- EAS:Friedel-Crafts Acylation Mechanism5m
- EAS:Any Carbocation Mechanism7m
- Electron Withdrawing Groups22m
- EAS:Ortho vs. Para Positions4m
- Acylation of Aniline9m
- Limitations of Friedel-Crafts Alkyation19m
- Advantages of Friedel-Crafts Acylation6m
- Blocking Groups - Sulfonic Acid12m
- EAS:Synergistic and Competitive Groups13m
- Side-Chain Halogenation6m
- Side-Chain Oxidation4m
- Reactions at Benzylic Positions31m
- Birch Reduction10m
- EAS:Sequence Groups4m
- EAS:Retrosynthesis29m
- Diazo Replacement Reactions6m
- Diazo Sequence Groups5m
- Diazo Retrosynthesis13m
- Nucleophilic Aromatic Substitution28m
- Benzyne16m
- 20. Phenols55m
- 21. Aldehydes and Ketones: Nucleophilic Addition4h 56m
- Naming Aldehydes8m
- Naming Ketones7m
- Oxidizing and Reducing Agents9m
- Oxidation of Alcohols28m
- Ozonolysis7m
- DIBAL5m
- Alkyne Hydration9m
- Nucleophilic Addition8m
- Cyanohydrin11m
- Organometallics on Ketones19m
- Overview of Nucleophilic Addition of Solvents13m
- Hydrates6m
- Hemiacetal9m
- Acetal12m
- Acetal Protecting Group16m
- Thioacetal6m
- Imine vs Enamine15m
- Addition of Amine Derivatives5m
- Wolff Kishner Reduction7m
- Baeyer-Villiger Oxidation39m
- Acid Chloride to Ketone7m
- Nitrile to Ketone9m
- Wittig Reaction18m
- Ketone and Aldehyde Synthesis Reactions14m
- 22. Carboxylic Acid Derivatives: NAS2h 51m
- Carboxylic Acid Derivatives7m
- Naming Carboxylic Acids9m
- Diacid Nomenclature6m
- Naming Esters5m
- Naming Nitriles3m
- Acid Chloride Nomenclature5m
- Naming Anhydrides7m
- Naming Amides5m
- Nucleophilic Acyl Substitution18m
- Carboxylic Acid to Acid Chloride6m
- Fischer Esterification5m
- Acid-Catalyzed Ester Hydrolysis4m
- Saponification3m
- Transesterification5m
- Lactones, Lactams and Cyclization Reactions10m
- Carboxylation5m
- Decarboxylation Mechanism14m
- Review of Nitriles46m
- 23. The Chemistry of Thioesters, Phophate Ester and Phosphate Anhydrides1h 10m
- 24. Enolate Chemistry: Reactions at the Alpha-Carbon1h 53m
- Tautomerization9m
- Tautomers of Dicarbonyl Compounds6m
- Enolate4m
- Acid-Catalyzed Alpha-Halogentation4m
- Base-Catalyzed Alpha-Halogentation3m
- Haloform Reaction8m
- Hell-Volhard-Zelinski Reaction3m
- Overview of Alpha-Alkylations and Acylations5m
- Enolate Alkylation and Acylation12m
- Enamine Alkylation and Acylation16m
- Beta-Dicarbonyl Synthesis Pathway7m
- Acetoacetic Ester Synthesis13m
- Malonic Ester Synthesis15m
- 25. Condensation Chemistry2h 9m
- 26. Amines1h 43m
- 27. Heterocycles2h 0m
- Nomenclature of Heterocycles15m
- Acid-Base Properties of Nitrogen Heterocycles10m
- Reactions of Pyrrole, Furan, and Thiophene13m
- Directing Effects in Substituted Pyrroles, Furans, and Thiophenes16m
- Addition Reactions of Furan8m
- EAS Reactions of Pyridine17m
- SNAr Reactions of Pyridine18m
- Side-Chain Reactions of Substituted Pyridines20m
- 28. Carbohydrates5h 53m
- Monosaccharide20m
- Monosaccharides - D and L Isomerism9m
- Monosaccharides - Drawing Fischer Projections18m
- Monosaccharides - Common Structures6m
- Monosaccharides - Forming Cyclic Hemiacetals12m
- Monosaccharides - Cyclization18m
- Monosaccharides - Haworth Projections13m
- Mutarotation11m
- Epimerization9m
- Monosaccharides - Aldose-Ketose Rearrangement8m
- Monosaccharides - Alkylation10m
- Monosaccharides - Acylation7m
- Glycoside6m
- Monosaccharides - N-Glycosides18m
- Monosaccharides - Reduction (Alditols)12m
- Monosaccharides - Weak Oxidation (Aldonic Acid)7m
- Reducing Sugars23m
- Monosaccharides - Strong Oxidation (Aldaric Acid)11m
- Monosaccharides - Oxidative Cleavage27m
- Monosaccharides - Osazones10m
- Monosaccharides - Kiliani-Fischer23m
- Monosaccharides - Wohl Degradation12m
- Monosaccharides - Ruff Degradation12m
- Disaccharide30m
- Polysaccharide11m
- 29. Amino Acids3h 20m
- Proteins and Amino Acids19m
- L and D Amino Acids14m
- Polar Amino Acids14m
- Amino Acid Chart18m
- Acid-Base Properties of Amino Acids33m
- Isoelectric Point14m
- Amino Acid Synthesis: HVZ Method12m
- Synthesis of Amino Acids: Acetamidomalonic Ester Synthesis16m
- Synthesis of Amino Acids: N-Phthalimidomalonic Ester Synthesis13m
- Synthesis of Amino Acids: Strecker Synthesis13m
- Reactions of Amino Acids: Esterification7m
- Reactions of Amino Acids: Acylation3m
- Reactions of Amino Acids: Hydrogenolysis6m
- Reactions of Amino Acids: Ninhydrin Test11m
- 30. Peptides and Proteins2h 42m
- Peptides12m
- Primary Protein Structure4m
- Secondary Protein Structure17m
- Tertiary Protein Structure11m
- Disulfide Bonds17m
- Quaternary Protein Structure10m
- Summary of Protein Structure7m
- Intro to Peptide Sequencing2m
- Peptide Sequencing: Partial Hydrolysis25m
- Peptide Sequencing: Partial Hydrolysis with Cyanogen Bromide7m
- Peptide Sequencing: Edman Degradation28m
- Merrifield Solid-Phase Peptide Synthesis18m
- 32. Lipids 2h 50m
- 34. Nucleic Acids1h 32m
- 35. Transition Metals5h 33m
- Electron Configuration of Elements45m
- Coordination Complexes20m
- Ligands24m
- Electron Counting10m
- The 18 and 16 Electron Rule13m
- Cross-Coupling General Reactions40m
- Heck Reaction40m
- Stille Reaction13m
- Suzuki Reaction25m
- Sonogashira Coupling Reaction17m
- Fukuyama Coupling Reaction15m
- Kumada Coupling Reaction13m
- Negishi Coupling Reaction16m
- Buchwald-Hartwig Amination Reaction19m
- Eglinton Reaction17m
Hydrogenation of Alkynes - Online Tutor, Practice Problems & Exam Prep
 Created using AI
Created using AIHydrogenation reactions convert triple bonds into more saturated forms, either double or single bonds. Full saturation uses catalytic hydrogenation or Wilkinson's catalyst, resulting in cis products through syn addition. Partial saturation includes dissolving metal reduction, yielding trans double bonds via anti addition, and Lindlar's catalyst, producing cis double bonds through syn addition. Understanding the stereochemistry and reagents is crucial for synthesizing specific molecular structures.
Hydrogenation reactions are a subtype of reduction reactions that add hydrogens to double and triple bonds. These differ in the types of products made and the stereochemistry of the mechanism.
The definition of hydrogenation.
Video transcript
Using Catalytic hydrogenation or Wilkinson’s Catalyst to turn alkynes to alkanes.
Video transcript
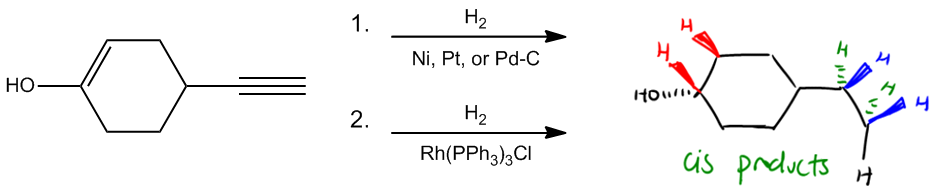
So let's look at the first and probably simplest form of hydrogenation, which we call full saturation. What that means is that I'm taking a double bond or a triple bond and I'm fully saturating it with hydrogens. Alright? We can do this using two different reagents. We can achieve this with either catalytic hydrogenation, which involves the reagents shown here in the first arrow. That would be basically H 2 over a plate of platinum, palladium, or nickel catalyst. Okay? Or there's another reaction we can use called Wilkinson's catalyst. And Wilkinson's catalyst is used for the same purpose: to take double bonds and triple bonds and fully saturate them to alkanes. Alright? As you can see, the reagents for Wilkinson's catalyst are a bit confusing. It's got the H 2 because we're adding hydrogen of course, but the catalyst is way different. In this case, we're going to use rhodium with three triphenylphosphines and then a chlorine. It looks messy. Okay? I'm not going to draw the whole thing for you as long as you can recognize that that's Wilkinson's, that's fine. Okay?
The only thing about full saturation is that this is a bit of a misnomer because remember that rings also count towards unsaturation. A ring is something that's missing two hydrogens because two ends are attached to each other. And actually, these reactions do nothing to rings. So what's going to happen is that all the double bonds and triple bonds will be gone, but the rings will stay intact. Alright?
One more thing before I draw these products. Notice that the stereochemistry mentions syn addition. Okay? Syn addition is another way to say that we're going to get cis products. So, what that means is that I would expect the hydrogens that we're adding to add from the same side of the double bond or the same side of the triple bond. So let's go ahead and draw our products. Basically, I would get the same exact sigma framework. When I say sigma framework, I'm just saying all the sigma bonds are the same. But now I would get a single bond over here, so that triple bond just turned into an alkane. And I would get that this alcohol is still here. And then I would finally get that the double bond is also gone, meaning that there are no pi bonds left. Okay? This is really good at getting rid of pi bonds. That's pretty much it.
Here I did include stereochemistry because there are no chiral centers, so I don't really have to. But if you're wondering where these hydrogens came from, especially maybe on the double bond, they could have both come from the same side. So that means let's say that one of the hydrogens came from here and one came from here. Notice that they came from the same side of the double bond, then that means that my alcohol will be forced downward. Alternatively, I could have also had the hydrogens add from the bottom, which means that the OH would have gone up as well. The same thing with the triple bond, except that for the triple bond, I added four hydrogens. I didn't just add two. So I basically added, let's say, two from the front, H 2 , and I also added two from the back, H 2 . So what I wound up getting was instead of, and then also, now this carbon here makes sense. It has CH2. But this one looks like it doesn't make sense because it's also CH2. But remember, there was already one H to begin with on that triple bond. So that last H is still there and it's right next to my head. So does that make sense? Basically, I added the two blue hydrogens, then I added two green hydrogens, and then this black H was just always there. Alright? So basically, you can think that full saturation, with either one of these two catalytic methods or Wilkinson's, is going to completely get rid of all pi bonds, all double bonds, and all triple bonds. Easy. Right? Cool.
Catalytic Hydrogenation or Wilkinson’s Catalyst: Alkane products
Stereochemistry: Syn Addition
Using Dissolving Metal Reduction or Lindlar’s Catalyst to turn alkynes to alkenes.
Video transcript
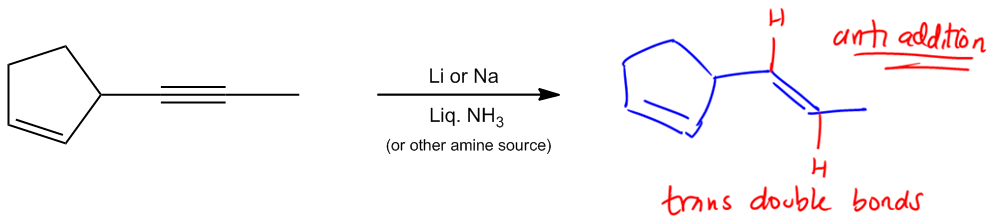
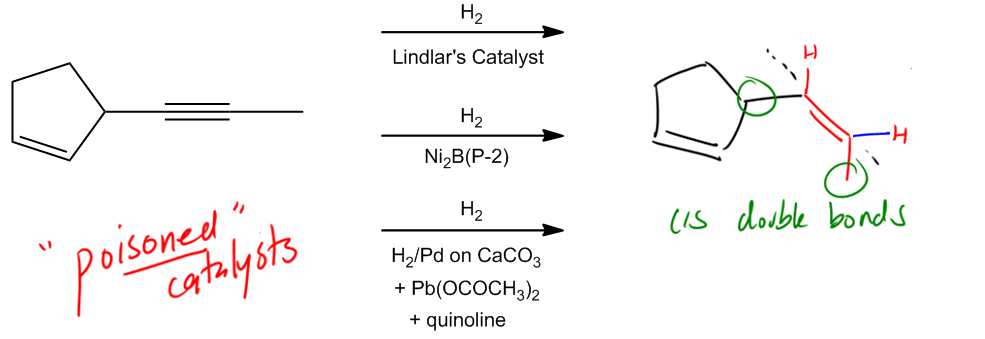
Move on to the next reaction. And actually, these next two are both considered partial saturation. So what that means is that for these next two, I'm not saturating all the way to an alkane. In fact, what I'm doing is I'm taking a triple bond and I'm only reacting it to a double bond. So I am adding two hydrogens, but I'm stopping there. Okay? And the first of these, really the biggest difference between them, other than the reagents, which are totally different, are the stereochemistry. Notice that my first one is anti addition. Notice that my second one is syn addition. That's going to have a difference in what my products look like. So let's look at this first one. The first one is called dissolving metal reduction. Okay? Dissolving metal reduction is just another name for these reagents. And what it is is that we have some kind of metal, lithium or sodium. Your professor can use either one in some kind of liquid amine source. Many times that amine is going to be NH3, but there's a lot of other things it could be. It could be NH2CH3 or it could be just any kind of usually primary or secondary amine. Alright? Cool. So we've got that part down. What does it do? Well, we learned I just taught you guys that partial saturation means that it's only working on triple bonds. So notice that I have the same or a very similar molecule here and what I'm wondering is how should I draw this? I have the same sigma framework, but for example, should I draw the double bond, or should I leave it there? I should leave it there because it turns out that since this is partial saturation, it has no effect on alkenes. No effect on alkenes. Alright? It's only going to work on triple bonds. So now I've got this triple bond. Is it going to work on this one? Absolutely. Well, what does anti addition mean? Anti addition, if you remember, that just means I have trans products. Okay? So what that means is that I would expect that my hydrogens are going to add from different sides of this double bond. What that means is that I should actually draw the double bond in a transposition. Why? Because the two hydrogens that I added must have added on different sides. Okay? Of different sides of the double bond, which means that they were anti addition. Is that making sense? So what that means is that dissolving metal reduction is important because this is a way that we can turn triple bonds into trans double bonds. That's going to be your product. Trans double bonds. Pretty cool, right? So that's something you have to keep in mind because, for synthesis later, when we start trying to make molecules, that's going to be the only way that we know how to turn a Sherbob wand and make it trans. Cool? So now let's go to this next one, which I'm sure you guys can already kind of anticipate what's going to happen. Notice that this one is called Lindlar's catalyst. And Lindlar's catalyst has a lot of different reagents that you could use. Unfortunately, different textbooks, different professors have their own way of writing it. Some professors are going to be really easy. They're going to be like, You know what? F all these reagents. We're just going to write Lindlar's. Okay? They're going to put H2 in Lindlar's, and you're going to know that that's Lindlar's. But some professors are going to use the actual reagents that are in the textbook, so some textbooks have this as the P2 catalyst. Okay? So the P2 catalyst is one form of Lindlar's. Another form is actually with, lead acetate, and quinoline. These are reagents that you just have to recognize that they are Lindlar's. You don't need to know exactly what they look like or draw them or even know the mechanism. But what you do need to know is to recognize that these are Lindlar's. These are also, just so you guys know, another term that you may hear is that these are poisoned catalysts. Okay? Now what could oops, Catalysts. Wow. Okay. Sorry, guys. Spelling isn't my forte. So poison catalyst. What does poisoned mean? What it means is that these catalysts have been created in such a way that they're not going to reduce all the way, or they're not going to hydrogenate all the way. Instead, they're going to stop at a given point.
Dissolving Metal Reduction: Alkene products
Stereochemistry: Anti Addition
Lindlar’s Catalyst: Alkene products
Stereochemistry: Syn Addition
Predict the product of the multistep synthesis





Do you want more practice?
More setsHere’s what students ask on this topic:
What is the difference between full saturation and partial saturation in hydrogenation of alkynes?
Full saturation in hydrogenation of alkynes involves converting a triple bond all the way to a single bond, resulting in an alkane. This is typically achieved using catalytic hydrogenation (H2 over Pt, Pd, or Ni) or Wilkinson's catalyst. The process results in syn addition, producing cis products. Partial saturation, on the other hand, stops at the double bond stage, converting a triple bond to a double bond. This can be done using dissolving metal reduction (yielding trans products via anti addition) or Lindlar's catalyst (yielding cis products via syn addition). Understanding these differences is crucial for synthesizing specific molecular structures.
 Created using AI
Created using AIWhat reagents are used in catalytic hydrogenation and Wilkinson's catalyst for full saturation of alkynes?
Catalytic hydrogenation for full saturation of alkynes typically uses H2 gas in the presence of a metal catalyst such as platinum (Pt), palladium (Pd), or nickel (Ni). Wilkinson's catalyst, another method for full saturation, involves H2 and a rhodium complex with three triphenylphosphines and a chlorine ligand. Both methods result in the complete hydrogenation of triple bonds to single bonds, producing alkanes with syn addition, meaning the added hydrogens come from the same side of the bond.
 Created using AI
Created using AIHow does dissolving metal reduction differ from Lindlar's catalyst in the hydrogenation of alkynes?
Dissolving metal reduction and Lindlar's catalyst are both used for partial saturation of alkynes, but they differ in stereochemistry and reagents. Dissolving metal reduction uses metals like lithium or sodium in liquid ammonia (NH3) and results in trans (anti addition) double bonds. Lindlar's catalyst, which can include reagents like lead acetate and quinoline, produces cis (syn addition) double bonds. These differences are crucial for synthesizing specific molecular structures with desired stereochemistry.
 Created using AI
Created using AIWhat is syn addition and anti addition in the context of hydrogenation of alkynes?
In the context of hydrogenation of alkynes, syn addition refers to the addition of hydrogen atoms to the same side of the double or triple bond, resulting in cis products. This is seen in full saturation using catalytic hydrogenation or Wilkinson's catalyst, and in partial saturation using Lindlar's catalyst. Anti addition, on the other hand, involves the addition of hydrogen atoms to opposite sides of the bond, resulting in trans products. This occurs in partial saturation using dissolving metal reduction. Understanding these concepts is essential for predicting the stereochemistry of the hydrogenation products.
 Created using AI
Created using AIWhy are Lindlar's catalysts referred to as poisoned catalysts?
Lindlar's catalysts are referred to as poisoned catalysts because they are designed to stop the hydrogenation process at the double bond stage, preventing full saturation to an alkane. This is achieved by modifying the catalyst with substances like lead acetate and quinoline, which inhibit further hydrogenation. As a result, Lindlar's catalysts produce cis double bonds through syn addition, making them useful for partial saturation of alkynes.
 Created using AI
Created using AIYour Organic Chemistry tutors
- Answer Problem 39 , parts a–h, using 2-butyne as the starting material instead of propyne. h. H2/Lindlar cata...
- Answer Problem 39 , parts a–h, using 2-butyne as the starting material instead of propyne. g. excess H2, Pd/C
- What are products of the following reactions? b.
- What are products of the following reactions? a.
- What stereoisomers are obtained from the following reactions? b. CH3CH2C☰CCH2CH3→1.H2/Lindlar catalyst 2. D2...
- What stereoisomers are obtained from the following reactions? a. CH3CH2C☰CCH2CH3→1.Na, NH3(liq), −78°C 2. D2...
- (•••) LOOKING AHEAD CHAPTER 10 The following reaction was recently reported to have been performed electrochem...
- Show how you would convert a. oct-3-yne to cis-oct-3-ene. b. pent-2-yne to trans-pent-2-ene.
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- (••) For the alkynes shows here, show the product(s) expected to form when treated under the following conditi...
- Predict the product of the following alkyne reductions. (a)
- Predict the product of the following alkyne reductions. (b)
- Predict the product of the following alkyne reductions. (c)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- Suggest alkynes that might be used to make the following trans-alkenes. (a)
- When sodium generates electrons in the presence of ammonia, these electrons persist in solution, giving the bl...
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Predict the product of the following hydrogenation reactions run with a poisoned catalyst. (a)
- Hydrogenation of which alkynes would produce the following cis-alkenes? (c)
- The following deprotonation step occurs during the trans reduction of an alkyne. Calculate K_eq for this react...
- Sodium amide, the base we use to deprotonate terminal alkynes, is synthesized by reducing ammonia via a mechan...
- Reduction of an alkyne using the Lindlar catalyst, a reaction presented in Section 10.6.2, produces only the c...
- Identify the product when each of the following reactions is performed on the triglyceride of linoleic acid (l...
- Hydrogenation of which alkynes would produce the following cis-alkenes?(b) <IMAGE>
- Long, polarized bonds are also reducible in the same way that the C―C π bond of an alkyne is. Show a mechanism...
- What is the major product of the reaction of 1 mol of propyne with each of the following reagents?g. excess H2...
- Describe the alkyne you should start with and the reagents you should use if you want to synthesizec. trans-2-...
- What reagents would you use for the following syntheses?c. hexane from 3-hexyne
- How can the following compounds be prepared using ethyne as the starting material?e. <IMAGE>
- Show how each of the following compounds can be prepared using the given starting material, any needed inorgan...
- How can the following compounds be prepared using ethyne as the starting material?f. <IMAGE>
- What are products of the following reactions?c. <IMAGE>d. <IMAGE>
- Show how you would convertc. cis-cyclodecene to trans-cyclodecene.
- What reaction would acetylene likely undergo if it were kept at 1500 °C for too long?
- Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or termi...
- Predict the product of the following reactions. (b)
- Describe the alkyne you should start with and the reagents you should use if you want to synthesize b. cis-2-...
- Describe the alkyne you should start with and the reagents you should use if you want to synthesize a. pentan...
- We have studied a number of pericyclic reactions previously. Draw the mechanism of the steps shown. The sectio...
- What alkene would you start with if you wanted to synthesize b. ethylcyclopentane?
- a. How many alkenes could you treat with H2, Pd/C to prepare methylcyclopentane? b. Which of the alkenes is t...
- What reagents would you use for the following syntheses? a. (Z)-3-hexene from 3-hexyne
- What reagents would you use for the following syntheses? b. (E)-3-hexene from 3-hexyne
- Predict the product of the following hydrogenation reactions. (b)
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (f) < of re...
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (i) < of re...
- Show how you would accomplish the following synthetic transformations. Show all intermediates. (g) < of re...

