Now we're going to talk about a way that we can make double bonds out of alcohols. And this is through a mechanism called acid catalyzed dehydration. Okay? So how does this work? Well, remember back to when I've talked about leaving groups in the past. Okay? And a leaving group is even a concept from acid-base chemistry. All it means is that it's something that really doesn't want to leave, something that really doesn't want to get a negative charge. So another word for leaving group is just conjugate base. Okay? So if you maybe don't really remember exactly what a leaving group is, just think conjugate base, back to acids and bases. And basically, alcohols are really bad leaving groups or they're really bad conjugate bases. They hate to become OH-. Why? Because OH- is actually a really strong base. Remember that you always want to go from stronger to weaker? If we're making OH- hydroxide, that's a really strong base. Okay? So this is not going to be very favored to just leave a molecule. But it turns out that there is one thing we can do to make alcohol a better leaving group and that is to use. And if we can use some kind of acid, we can actually convert alcohol into an awesome leaving group. That awesome leaving group would just be that we add an H to it, we protonate it, so it turns into water. And water is an awesome leaving group because it's neutral. It loves to leave and it loves to just be by itself in solution. Okay? So here's the general formula. I'm not going to show you the mechanism just yet. But basically what we have is we have some kind of alcohol and we have some kind of acid over water. Now in this case, I just put the general H8. I mean, really any acid, but your common acids are going to be sorry sorry about that. Any of the strong acids. So H2SO4 is seen all the time. Any of the hydrogen halide strong acids. I've also seen phosphoric acid. This is not really a strong acid, but it's still strong enough to make the reaction go. These are all very frequently used acids for acid catalyzed dehydration. Okay? And what we're basically doing is we're taking an alcohol. We're going to be removing 2 sigma bonds. We're going to be removing the alcohol and a beta hydrogen. Okay. So this is my alpha. This is my beta carbon. Alpha and beta relative to the alcohol. We're going to be taking away 2 different sigma bonds and we're going to be making 1 pi bond instead. Now that general reaction of taking away 2 sigmas and making 1 pi is an elimination. So this is actually going to be an elimination reaction. Okay? Hope that's making sense so far. Now there's another reaction that you may have already learned or that you will learn soon and that's actually the opposite. It's called acid catalyzed hydration. Acid catalyzed hydration is a reaction where we go from the double bond and we go back to the alcohol. Okay? This is actually what's called an addition reaction. Okay? So if you remember back to the general types of reactions we talked about, that when you take one bond and you make it into 2, that's actually addition. When you take 2 bonds and you make it into 1, that's elimination. So hopefully that makes sense. So basically, the addition part is gonna be the hydration. The elimination part is the dehydration. Okay? How do you know which one it is? How do you know if it's going to be a hydration or a dehydration? You just look at what you're starting with. So in this case, since I have my alcohol, I know that I'm starting with an alcohol and I'm going to try to eliminate it with an acid to become a double bond. However, if I was starting with a double bond and I used acid, then I could add water to it and that would become a hydration. In this step, in this video, we're just gonna learn about dehydration, but I'm just asking you to keep this in the back of your mind later on for when you have to do practice problems with hydration. That this is the way that we tell the two reactions apart. Okay? So let's just talk more in-depth about dehydration. There's a few facts I want you to know. First of all, the more R groups on that alcohol, the easier it's gonna be to dehydrate. This is just a fact that might come up on maybe a conceptual part of your exam or your professor might even ask you, give you 4 different alcohols. Which one's the easiest to dehydrate? Tertiary would be the easiest. Secondary, primary, and then is the worst and then, Oops, sorry. I don't know what I'm trying to draw here. But basically, the methyl can't even happen because if it's a methyl alcohol then or methanol, that can't even eliminate cause it's only got one carbon. Okay? So basically, the easiest one is tertiary, the worst one is primary. That's the first thing. The second thing is that the specific elimination mechanism that we use is going to depend on how easily the molecule is going to form a carbocation. Okay? And the understanding of carbocations is kind of essential to these two mechanisms. All that means is that if you guys remember back to the trend of carbocation stability, that tertiary carbocations are the most stable and primary carbocations are the worst. Okay? And then obviously, methyl is even worse than that, but like I said, methyl doesn't even get counted because we can't use it.
Dehydration Reaction - Online Tutor, Practice Problems & Exam Prep
Dehydration reactions eliminate alcohols, yielding double bonds.
General features of acid-catalyzed dehydration.
Video transcript
Recall that for elimination to take place, you need a good leaving group. Alcohols are terrible leaving groups, but in the presence of acid, they can be converted into water, which is an amazing leaving group.

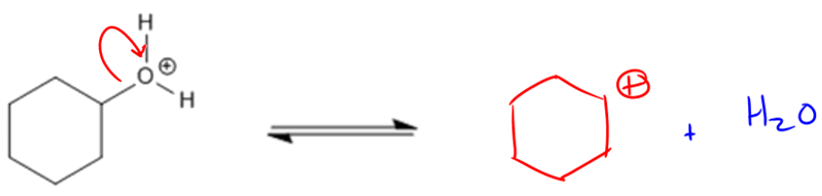
If an alcohol can form a stable carbocation, the E1 mechanism will be favored. If it can’t, then the mechanism will follow an E2 pathway. Let’s start off 1° ROH, which usually follow E2.
Dehydration of 1° alcohols:The E2 Mechanism
Protonation:
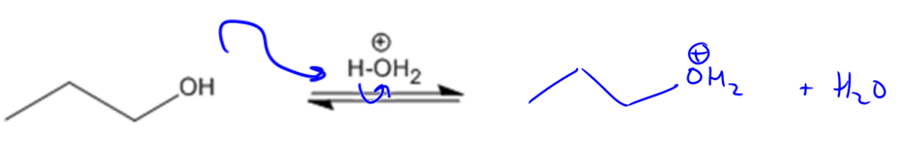
E2 Concerted β-Elimination:

Dehydration of 2° and 3° alcohols:The E1 Mechanism.
Protonation:

Formation of a Carbocation (Slow Step):
E1 β-Elimination (Fast Step):

An extra note of caution with 1° alcohols.
Video transcript
Let's do some cumulative practice based on everything we've learned from acid-catalyzed dehydration and make sure to be mindful of all the different details I taught you about which mechanism you would use and what would happen in terms of how many steps they would have and stuff like that. Now there is one instruction that I want to give you, and that's remember that I told you that primaries would do an E2 reaction or mechanism, and then secondaries and tertiaries would perform an E1. Okay? Now that is almost always true, but there is one exception to that. That is going to be if you have a primary alcohol that can rearrange to a tertiary carbocation through a shift, then it's actually going to do a carbocation mediated E1 mechanism instead. Okay? So you're going to have to use that information to maybe determine this first one. Notice that it is primary. Figure out, okay, this would usually be E2. If it could shift to a tertiary position, then you should actually use E1. But I'm going to let you guys figure that out.
The second one, same thing. You have to figure out what mechanism and everything that would happen in between. Alright? So go ahead and get started on this first one and then I'll show you guys how to do the second.
Remember how I mentioned that 1° alcohols usually follow E2?
This isn’t the case of 1° alcohols that can rearrange to 3° alcohols. Since the 1,2-rearrangement creates a super stable carbocation, the reaction will follow the E1 pathway.
Predict the major product of the reaction




Predict the major product of the reaction




Do you want more practice?
More setsYour Organic Chemistry tutors
- E1 eliminations of alkyl halides are rarely useful for synthetic purposes because they give mixtures of substi...
- Predict the products of the following reactions. When more than one product is expected, predict which will be...
- Predict the major products of acid-catalyzed dehydration of the following alcohols. (a) pentan-2-ol (b) 1-...
- Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only nee...
- Show the product(s) you expect from dehydration of the following alcohols when they are heated in sulfuric or ...
- Propose mechanisms for the following reactions. (d) H2SO4/H2O, heat—> (an interesting minor product)
- Propose mechanisms for the following reactions. (c) 2-methylclyclohexanol H2SO4, heat—> 1-methylcyclohexe...
- Propose mechanisms for the following reactions. (b) pentan-2-ol H2SO4, heat—> pent-1-ene + pent-2-ene (...
- Propose mechanisms for the following reactions. (a) cyclopentanol H2SO4, heat—> cyclopentene
- Show the product(s) you expect from dehydration of the following alcohols when they are heated in sulfuric or ...
- (•••) THINKING AHEAD The acid-catalyzed hydration we learned here in Chapter 8 is reversible [we introduce thi...
- (•••) THINKING AHEAD The acid-catalyzed hydration we learned here in Chapter 8 is reversible [we introduce thi...
- (•••) THINKING AHEAD The acid-catalyzed hydration we learned here in Chapter 8 is reversible [we introduce thi...
- If the compound shown in the margin is heated in the presence of H2SO4, a. what constitutional isomer would b...
- Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the fol...
- Explain why the acid-catalyzed dehydration of an alcohol is a reversible reaction, whereas the base-promoted d...
- What stereoisomers are formed in the following reactions? Which stereoisomer is the major product? a. the aci...
- Explain why the following alcohols, when heated with acid, form the same alkene.
- When the following seven-membered ring alcohol is dehydrated, three alkenes are formed. Propose a mechanism fo...
- When heated with H2SO4, both 3,3-dimethyl-2-butanol and 2,3-dimethyl-2-butanol are dehydrated to form 2,3-dime...
- Write the appropriate reagent over each arrow.
- What is the major product(s) of each of the following reactions? d.
- What is the major product obtained when each of the following alcohols is heated in the presence of H2SO4? b....
- What is the major product obtained when each of the following alcohols is heated in the presence of H2SO4? a....
- Propose a mechanism for each of the following reactions: a.
- Predict the major products of dehydration catalyzed by sulfuric acid. (a) hexan-1-ol (b) hexan-2-ol (c) pent...
- Identify the alcohol(s) that would produce the following alkenes under the given conditions. (a)
- When HCl was used for the attempted dehydration reaction shown, a reaction occurred, but none of the desired p...
- Predict the product(s) of the reactions shown. (b)
- Identify two different alcohols that can be dehydrated (one with rearrangement) to form the alkene shown.
- Identify two different alcohols that can be dehydrated (one without rearrangement) to form the alkene shown.
- (•••) Produce a mechanism for the following transformation.
- Identify the alcohol(s) that would produce the following alkenes under the given conditions.(b) <IMAGE>
- Identify the alcohol(s) that would produce the following alkenes under the given conditions.(c) <IMAGE>
- (•••) When using sulfuric acid, but in the absence of other nucleophiles like water or bromide ion, less stabl...
- Predict the product of the following pinacol rearrangements.(c) <IMAGE>
- (••••) Suggest a mechanism for the following reactions.(c) Elimination : ...
- (•••) Suggest a mechanism for the following substitution reaction. [We studied a similar reaction of radicals ...
- (••) Predict the product(s) that would result when molecules (a)–(p) are allowed to react under the following ...
- (•••) THINKING AHEAD The acid-catalyzed hydration we learned here in Chapter 8 is reversible [we introduce thi...
- What product is obtained when the following vicinal diol is heated in an acidic solution?<IMAGE>
- What stereoisomers are formed in the following reactions? Which stereoisomer is the major product?b. the acid-...
- Indicate which alcohol in each pair undergoes an elimination reaction more rapidly when heated with H2SO4.a. &...
- Explain why (S)-2-butanol forms a racemic mixture when it is heated in sulfuric acid.
- Propose a mechanism for the following reaction:<IMAGE>
- What alkenes would you expect to be obtained from the acid-catalyzed dehydration of 1-hexanol?
- Indicate which alcohol in each pair undergoes an elimination reaction more rapidly when heated with H2SO4.e. &...
- Indicate which alcohol in each pair undergoes an elimination reaction more rapidly when heated with H2SO4.d. &...
- Indicate which alcohol in each pair undergoes an elimination reaction more rapidly when heated with H2SO4.c. &...
- Propose mechanisms for the following reactions.HINT: Alcohol dehydrations usually go through E1 elimination of...
- Propose mechanisms for the following reactions.HINT: Alcohol dehydrations usually go through E1 elimination of...
- When the following substituted cycloheptanol undergoesdehydration, one of the minor products has undergonea ri...
- Propose a mechanism for each reaction.(c) <IMAGE of reaction>
- Propose a mechanism for each reaction.(a) <IMAGE of reaction>
- Propose mechanisms for the following reactions. In most cases,more products are formed than are shown here. Yo...
- Write a balanced equation for each reaction, showing the major product you expect.(a) CH3-CH2-CH(CH3)OH H2SO...
- Write a balanced equation for each reaction, showing the major product you expect.(c) <IMAGE> H2SO4, h...
- Show the product(s) you expect from dehydration of the following alcohols when they are heated in sulfuric or ...
- Write a mechanism that explains the formation of the following product. In your mechanism, explain the cause o...
- Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one ...
- Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only nee...
- Four pairs of compounds are shown. In each pair, one ofthe compounds reacts more quickly, or with a more favor...
- Predict the product of the following pinacol rearrangements. (b)
- Propose a mechanism for each of the following reactions: b.
- Propose a mechanism for each of the following reactions: a.
- The following reaction is called the pinacol rearrangement. The reaction begins with an acid-promoted ionizati...
- Treatment of the following alcohol was expected to give alkene A. Instead, B was produced as the major product...
- Although 2-methyl-1,2-propanediol is an unsymmetrical vicinal diol, only one product is obtained when it is de...
- A student was studying terpene synthesis, and she wanted to make the compound shown here. First she converted ...
- b. What is the product of the following reaction?
- Which of the following alcohols dehydrates the fastest when heated with acid?
- Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only nee...
- Predict the major products of acid-catalyzed dehydration of the following alcohols.(c) 2-methylcyclohexanol(d)...
- A vicinal diol has OH groups on adjacent carbons. The dehydration of a vicinal diol is accompanied by a rearra...
- Propose mechanisms for the following reactions. In most cases,more products are formed than are shown here. Yo...

