Now I want to talk about an analytical technique that's used to measure the amount of energy in a molecule. Alright? And that technique is called heat of combustion. Alright? So the heat of combustion is, like I said, basically a machine that blows up molecules to see how energetic they are. And basically, the theory behind it is this: the higher the heat of combustion, or basically the more heat that's released by the explosion, the higher the energy of the molecule. And if the energy is very high, that means we would expect it not to be very stable. Okay? Vice versa, the same thing would be if you have a low heat of combustion, then you have low energy in the molecule, which means it must be a very stable molecule. Alright? So that's really all we need to know for right now, but I just want you to understand the relationship here because your professor could use any word he wants. He could say, pick the most stable molecule or pick the most energetic molecule or pick the one with the lowest heat of combustion. You need to understand what all three of those things mean and how they relate to each other. Alright? So just think that heat of combustion releases energy, so those should be in the same direction, and stability should be the opposite of whatever those are.
Ring Strain - Online Tutor, Practice Problems & Exam Prep
We can use an instrument called a calorimeter to determine how much potential energy is stored in molecules.
The Relationship Between Heat of Combustion and Stability
Understanding Heat of Combustion
Video transcript

Factors that Affect Alkane Stability
Shape and strain make alkanes unstable
Video transcript
So now there are actually two different ways that alkanes can become unstable. The first one, and probably the most difficult to explain, is the shape. There's just this rule in organic chemistry that a straight chain is going to be less stable than a branched chain. Where does this come from? Actually, this is a topic of hot debate and there's actually research going on right now to figure out why straight chains are less stable than branched chains. It's not something that's talked about in your textbook very much, and all I would say is it has to do with very, very complicated things. It has to do with stuff that's well beyond the scope of this course. So what I would say is instead of trying to understand it, just memorize it. That a straight chain, if I had a 6-carbon straight chain, and if I had a 6-carbon branched chain, okay, we're not talking about intermolecular forces. Intermolecular forces, if you're thinking of van der Waals forces, that's something completely different. Okay? That has to do with what state of matter it's in. I'm talking about actually how energetic it is. Like how much energy it releases when I burn it. That is going to be higher for the straight chain and that's going to be lower for the branched chain. Why? Because the branched chain is going to be more stable, the straight chain is going to be less stable. That's something you should know. The second thing is strain. There's actually a lot of different types of strain. I'm going to talk about two right now, but there's actually even more than that.
Strain is a super general word. So now let’s go more into specifics of the types of strain you need to recognize.
Types of Strain
- Angle Strain
What is angle strain?
Video transcript
So, a lot of these are found in cycloalkanes. So the first one is angle strain. Okay? Angle strain exists when tetrahedral bonds are forced out of their ideal bond angle of 109.5° degrees. Remember that tetrahedrals always want to have 109.5°, and the smaller your rings get, the more difficult it is for those carbon-carbon bonds to maintain that kind of bonding. So, let me just give you an example of a triangle. I'm not sure if you guys remember from geometry, but a triangle, like if you have an equilateral triangle, each of these bonds or each of these corners is going to be 60° degrees. Okay? Is 60° close to 109.5°? Not even close. Remember that all of these carbons have two hydrogens. So what that means is that they are tetrahedrals and they want to have 109.5° bond angles, but they don't because of that stupid triangle. So what that means is that 60 is far less than 109.5, so this is going to be highly strained. And if it's highly strained, guess what that means? That means it's going to have high energy and if it has high energy, then you can guess the rest. That means it has low stability. Alright? So let's keep going. The square is a little bit better, actually a lot better. It's at 90° degrees, but that's still pretty far off from 109.5°. Alright, so in this case, this one's a little bit better, but still, it still has angle strain. Now, a 5-membered ring is getting a lot closer to what we want. A 5-membered ring will have bond angles of 108°. And 108° is almost the same as 109.5°, like there's almost no difference there. So in terms of angle strain, a 5-membered ring, cyclopentane has very little angle strain. Are you guys cool with that? It's almost perfect. Then let's look at finally a 6-membered ring. A 6-membered ring would actually have bond angles of 120° degrees if it were drawn in a planar form. Well, 120° degrees is way more than 109.5°, so I would expect that cyclohexane would actually be less stable than cyclopentane. But actually, it turns out to not be true. Cyclohexane actually turns out to be the most stable ring out of all the rings. Okay? You can make as many carbons, you can make tons of carbons, but cyclohexane is the most stable ring. Why is that? That if the bond angle is so off, oops, I didn't mean to do that. I meant to highlight it. If the bond angle is 120°, then why would it be the most stable? Okay? And we're going to learn that in a little bit.
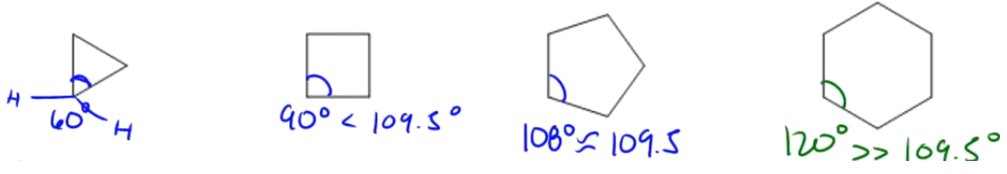
The ideal bond angle for sp3 hybirdized carbon is 109.5°, so the more we deviate from that number, the more unstable the angle will be! (Aka cyclopropane sucks).
- Torsional Strain
What is torsional strain?
Video transcript
Then we have Torsional strain. Torsional strain is a different type of strain that exists when carbons, which have hydrogens attached, overlap in space. Basically, the hydrogens will be eclipsed in space. So, let me give you another example of a cyclobutane here. This is just the square, but now we're looking at it in a 3D version. Later on, I'm going to teach you what the name of that is, but don't worry about it for right now. And what this means is all of these hydrogens are facing the same exact direction. They're all looking at each other exactly the same way. So that means that they're all eclipsed over each other. For example, if I had an eyeball looking here, I would see that all of these are eclipsed and then all the red ones are eclipsed. That is called torsional strain. Torsional strain is the strain that comes from having eclipsed bonds. Now you can see that cyclopentane is a little bit better, but it still has some eclipsed bonds here and here. Usually, cyclopentane will kind of move out of the plane, like it will bend a little bit, so that it won't have so many eclipsed bonds. But it turns out that if your professor were to ask you what is the main source of instability for cyclopentane? By the way, this is just a 3D version of that. Is the main cause of the strain, oh, I'm sorry, the main cause of instability, is it ring strain, which is right over there? Or is it torsional strain, which is those hydrogens there? The answer is that it's actually torsional strain. Torsional strain is the reason that cyclopentane isn't very stable, or isn't as stable because of the fact that no matter how much it folds, it's always going to have some overlapping hydrogen atoms here and some overlapping hydrogen atoms here. And that means that they're going to be in an eclipsed conformation and they're going to be kind of running into each other and bumping into each other and that's not very good.
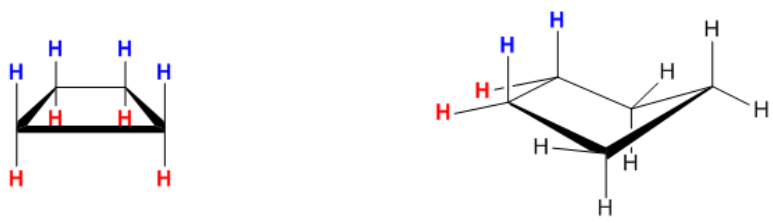
Torsional strain increases with the number of eclipsing hydrogens in a molecule. Some of these rings are so small they can’t twist to prevent these interactions, which makes them unstable.
Lowest Heat of Combustion
Video transcript
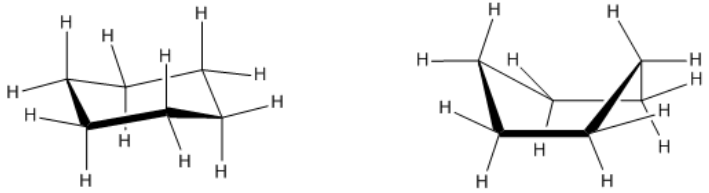
What I want to do is show you 2 3D versions of cyclohexane, and I want you guys to tell me which one you think is going to have the lowest heat of combustion. Alright? I haven't even taught you about cyclohexane yet, but I just want you guys to predict which one has the lowest heat of combustion. So go ahead and just like pause the video, and then when you're done thinking, also think: What does the lowest heat of combustion mean? So once you're done thinking, unpause the video.
Alright, so I'm pretty sure that you guys got that the lowest heat of combustion means the lowest energy and that lowest energy means most stable. Okay? So we're looking for the one that has the least amount of strain. What we found is that essentially this one over here would actually have quite a bit of torsional strain. Why? Because check it out, I have these hydrogens that are poking at each other. They're basically running into each other's space. So that would be one source of strain. Another source of strain would be that you have these hydrogens down here that are pretty much in each other's way. Okay? They're also eclipsed. Okay? So this would not be the best conformation for cyclohexane.
Now, over here on this one, this one is way better because we actually don't have any direct torsional strain. Now you might be wondering, well Johnny, I see that we have these hydrogens here that are facing the same way, but they have a carbon in between. That means they're actually pretty far apart from each other. Okay? There's actually very little torsional strain on this kind of conformation. Okay. And I wanted to tell you guys that it turns out that this is what cyclohexane is actually going to look like in real-life. In real life, instead of having 120 degree bond angles and having everything be eclipsed, all the hydrogens be eclipsed, instead what it does is it forms a puckered conformation and turns into what we call a chair.
And later on, when I talk about cyclohexane, and they have almost no torsional strain, and they have almost no torsional strain. So they are like the ideal cycloalkanes because they have pretty much the perfect bond angle, and because they are all twisted in that chair conformation, there's no torsional strain or very little torsional strain.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Cyclopropane (C3H6, a three-membered ring) is more reactive than most other cycloalkanes. c. Suggest why cycl...
- The heat of combustion of cis-1,2-dimethylcyclopropane is larger than that of the trans isomer. Which isomer i...
- Verify the strain energy shown in Table 3.8 for cycloheptane
- Which conformation in each of the following pairs has the least strain energy?(b) <IMAGE>
- Which conformation in each of the following pairs has the least strain energy?(a) <IMAGE>
- Choose the conformation in each pair that is most stable. If both are equally stable, then write 'no differenc...
- (••••) The normal C(sp³) - C (sp³) bond length is 1.54 Å. The normal bond angle for an sp³-hybridized carbon ...

