So now we're just going to start layering stuff onto these alkanes and making the names more complex. Let's talk about what happens when you have a ringed structure. So cycloalkanes are the name given to any time that you have a ring inside of your alkane. And we're going to start off with the easy ones, which are just monocyclic compounds. Monocyclic just means one ring. Okay. And these are easy. All we're going to do is we're just going to attach cyclo to the beginning of the root chain. Okay? So all of a sudden hexane becomes cyclohexane. Alright? If it's a ring. The root is assigned to the portion of the alkane with the greater number of carbons. Now where this comes into play is that usually it's really obvious which one is bigger or which one is going to get the root name. But sometimes it's not as obvious, meaning that sometimes you have both a long chain and a ring on the same structure. Most of the time it's just going to be either a chain or it's going to be a ring. But sometimes some structures combine both. What do we do if we combine both? Okay. Well, then what we want to do is we want to give the part with the greater number of chains the root.
Naming Cycloalkanes - Online Tutor, Practice Problems & Exam Prep
Ringed structures are easy to name, you just need to use a new prefix (aka –cyclo)!
Hint: Benzene and a cyclohexane are NOT the same thing. Remember, benzene has double bonds in it!
Understanding Cycloalkanes
How to find the root name for cycloalkanes
Video transcript
In general, we assign the root name to the portion of the alkane that has the greater number of carbons.
Determining Root Name
Video transcript
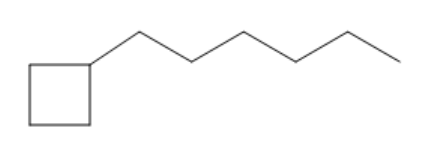
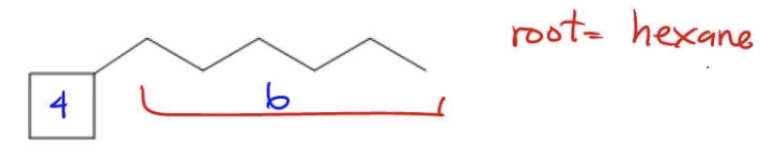
So let's just go down to this example for a second. We're not going to finish it, but I just want you guys to tell me which one would be the root. Here I have a 4 carbon ring and I have a 6 carbon chain. Here I have a 6 carbon ring and a 4 carbon chain. So in example a, which one would be the name of the root carbon? Well, the root carbon is chain, is a chain. It's these 6 carbons here because it's the largest portion. So the root in this case would just equal hexane. Does that make sense? Because it's just a 6 carbon chain.
Determining Root Name
Video transcript
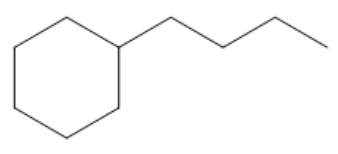
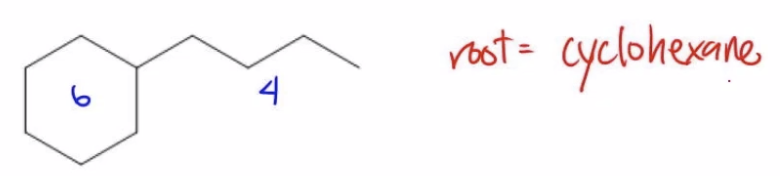
Now I look at example b. Example b also has a 6 carbon portion, but that's in the ring, and that's the greater portion. So in this case, the root is actually going to be cyclohexane. Does that make sense? Because now my ring is the part that's getting the root name. I hope that makes sense.
Numbering Monocyclic Cycloalkanes
If you only have one substituent on your ring, the numerical location is unnecessary!
Why it is okay to omit a single location for monocyclics
Video transcript
Then lastly, if there's only one substituent on your ring, so let's say you have a ring and you have one thing coming off of it. The location of that thing can be omitted. How does that make sense? Well, because if you have a chain, let me give you an example chain. Okay? Obviously, this is the ugliest chain ever. I didn't even do the zigzags. But if you have a chain and you add one thing to it, that thing could be in a lot of different places. It could be there, or I could erase it and I could put it there, or I could erase it and I could put it right at the end. Those are all different possibilities of where that stick could be. Do you just see how I'm saying that the location is going to matter? That is a different location than that. But if I have a ring and I put it here, that's the same thing as if I put it here, and that's the same thing as if I put it here. All of them are the same because of the ring, I can rotate as much as I want, whereas the chain, if I put it in the middle, it's stuck in the middle. It's never going to go to the end. Does that kind of make sense? So for a chain, you always have to say the location. Always note the location. But for a ring, the location can be omitted. Does that make sense? Now this is only true if I have one group. If I have more than one branch, so let's say I have 2 branches, now you need to say what the locations are. Why? Because that is going to be a very different structure than that, and that's going to be a different structure than that. So then, once I have 2 things, it breaks that rule. Remember, I'm just trying to say, if you only have one thing coming off of your ring, many times that location will be omitted. Does that make sense? Cool.
Time to complete those names. Let's give it a try.
Name the following alkane
Video transcript
So here's the substituent. Here's the substituent. So why don't you tell me the first one. What would be the name of that substituent? And if location is important, tell me what the location would be as well. So first of all, what is 4? I mean, what is this 4 carbon thing? It's a cyclobutane. Do you guys agree with that? Cyclobutane. But is it the main chain? Is it the main root? No, it's not. It's a substituent. So remember, there's always an ending that we give every substituent. What is that? Substituents always get a -yl ending. So what that means is that this is actually going to be called cyclobutyl. The reason it's cyclobutyl is because it's not the root, it's a substituent. Does that make sense? Then what's the location? The location would just be that it's on the first carbon of this, so it would be one cyclobutyl because on my 6 carbon chain, that's going to be the side that I want to have the lowest number. It would be dumb for me to put this side as the lowest number because then it would take me 6 carbons to get to the cyclobutyl. So there we go. This name is going to be 1-cyclobutylhexane. Makes sense? Sorry, my handwriting got really messy there. I'll try to make it better.
Name the following alkane
Video transcript
So for this one, what type of substituent do I have? Well, for this one, I actually just have a four-carbon chain. So the name for that would be just butyl. Does that butyl have a location? Technically, if I want to give it a location, I could give it the location 1 because obviously, I'm going to start numbering the ring wherever the first substituent is because I can number the ring from wherever. But since there's only one substituent on here, I could also just drop the 1 and I could just call this butylcyclohexane. And what that tells the reader is that hey, I have a ring and I have one thing coming off of it. It doesn't matter where you write that butyl group because they're all the same no matter what. Cool? Awesome. So now we know how to name cycloalkanes.
Great job! Did you remember to include the location for the first example? Remember, that location is not optional!
Introduction to Bicyclics
Bicyclics are also forms of cycloalkanes, but since they are not monocyclic, they have completely different rules for naming! (See next topic)
What is a bicyclic molecule?
Video transcript
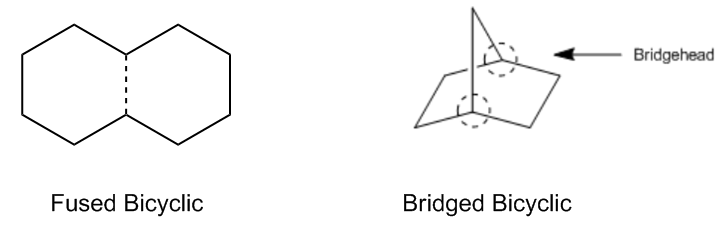
Now I just want to introduce bicyclics, and I'm not going to rigorously teach you how to name them here. In fact, I'm not going to teach you how to name them unless your professor specifically asks. Because bicyclics are kind of like iffy. Some professors want you to know them, some professors don't. But I'm just going to teach you, no matter what, you should know the basics of what a bicyclic is. And bicyclics are composed of 2 distinct rings attached along one bond. So, this would be an example of a bicyclic and it's made out of 2 cyclohexanes. And the actual name for a bicyclic of 2 cyclohexanes is called a decalin. So decalin just equals cyclohexane bicyclic. Some professors also take a special interest in decalins, and I will also be monitoring your class to see if I have to teach a separate section on decalins as well. Some professors don't really care. What's important is that I just want you to know that a bicyclic, by the way, this dotted bond here is the same thing as a regular bond. I'm just pointing out that this is the bond that's shared.
Now a bridged compound is a type of bicyclic and it's actually composed of 3 compound rings attached by what we call 2 bridgeheads. I know this is getting a little weird. Here's an example. This one's called Norbornene and it's a very common, it's actually one of the most common bridge structures. And you're asking me, "Johnny, where are the 3 rings?" Well, there actually are. There's this main thing down here that's actually just a weird way to draw cyclohexane. So that's just a 6-membered ring. Then I've got a 5-membered ring if I go along one side and then up like that. So that's one 5-membered ring. And then it turns out that I have another 5-membered ring if I go up the other side and up that thing. This thing in the middle that I keep pointing to is called the bridge. So it's like, I don't know, think about that you're walking over a bridge and you're going from one side of the molecule to the other. The atoms that attach all of those are called the bridgehead atoms. So that's what I meant by 2 bridgeheads. So this is called a bridged compound. So these are going to get into different ways of naming bicyclics. These have a certain way of naming, but like I said, that's going to be a separate section that I teach you only if your professor requires that you know that. I just want you to be familiar with what a bridge is.
Awesome guys. So with that said, let's go ahead and move on to the next topic.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Each of the following descriptions applies to more than one alkane. In each case, draw and name two structures...
- b. Draw and name the six isomeric cyclopentanes of molecular formula C7H14. These will include four constituti...
- a. Draw and name the five cycloalkane structures of formula C5H10. Can any of these structures give rise to ge...
- trans-1,2-Dimethylcyclobutane is more stable than cis-1,2-dimethylcyclobutane, but cis-1,3-dimethylcyclobutane...
- Name the following cycloalkanes using the IUPAC system of nomenclature. [Hint: Each molecule exemplifies one o...
- Name the following cycloalkanes using the IUPAC system of nomenclature. [Hint: Each molecule exemplifies one o...
- Name the following cycloalkanes using the IUPAC system of nomenclature. [Hint: Each molecule exemplifies one o...
- Name the following cycloalkanes using the IUPAC system of nomenclature. [Hint: Each molecule exemplifies one o...
- Give IUPAC names for the following compounds. a. b. c.
- Classify each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°). c. ...
- (•••) Correct the following incorrect names using standard IUPAC nomenclature. [Draw a compound that correspon...
- Each of the following descriptions applies to more than one alkane. In each case, draw and name two structures...
- (•••) Correct the following incorrect names using standard IUPAC nomenclature. [Draw a compound that correspon...
- Draw the structure and give the molecular formula for each of the following compounds. a. 1-ethyl-3-methylcyc...
- Draw the structure that corresponds with each name. e. 2,2,4,4-tetramethylhexane f. trans-1,3-diethylcyclopen...
- What is each compound's systematic name? e. f.
- (•••) Looking down the indicated bond, show the three most stable conformations and choose the one that is mos...
- a. Draw all the isomers with molecular formula C6H12 that contain a cyclobutane ring.

