Now that we understand the positions of cyclohexane, I'm actually going to take like 5 minutes just to teach you how to draw it. I know that sounds kind of juvenile, but once again, there are so many people that struggle to draw this weird shape that it's not even worth it not to go over it. We should just go over it once, make sure that you have at least one solid way how to draw it, and then we'll move on from there. Okay? So how to draw a cyclohexane? The easiest I've actually seen a lot of different explanations in different books. The easiest one that I've found is this. I always just draw 2 slightly angled parallel lines like that. Okay? And then all I do is I cap off both ends. What that means is I use an up cap for the top one and I use a down cap for the bottom one. Okay? That one actually came out really nice. You think that I'm a pro. Well, I kind of am, but they usually come out not that nice. So that's how to draw a cyclohexane. And it turns out that if you want to draw the other one, you should be able to draw both. You would draw the slightly angled parallel lines the other way, and then you'd cap them the other way. Okay, so that one came out okay too. Okay? These are totally legit. This would totally work in a test. Alright? I'm sure that some of you girls can draw this way nicer than I can. But whatever. I'm just saying this works.
Cis vs Trans Conformations - Online Tutor, Practice Problems & Exam Prep
Before we can really understand chair conformations, we have to practice drawing them! These tricky little suckers can be hard to get right.
Method for Drawing Chairs
How to draw chairs.
Video transcript
- Draw two slightly angled parallel lines.

- Cap both ends off

How did you do?
It's ok if you didn't draw it beautifully the first time - practice makes perfect!
Cis and Trans
How to distinguish cis from trans.
Video transcript
I want to teach you guys how to determine cis and trans. Okay? Cis and trans are not based on whether your positions are axial or equatorial. That's one of the biggest misconceptions in this chapter. A lot of students say, "oh, but they're both equatorial, so that means that they should be cis." Or "one is cis and one is equatorial and one is axial so they should be trans." Wrong. That's not how you decide it at all. And trans is actually going to be based on whether the groups are facing the same face of the ring. What I mean by face is just top or bottom. Okay? So what that means is that I'm not going to be looking at positions. I'm going to be looking at direction.
Cis or trans is based on whether the groups are facing the same face (top or bottom) of the ring.
- It has nothing to do with the axial and equatorial positions!
Is the following cyclohexane cis or trans?
Video transcript
Overall, I have an axial position. This would be axial, right? And then this would be an equatorial position. Do you agree with me so far? Because one is going straight up, and then one is going in a slightly opposite direction of the axial that would be there. Okay? But that's not what I care about when I think about persistent trans. All I think about is what direction it is facing on the ring. Well, this one is facing the top part of the ring, and this one would also be facing the top part of the ring because it's going slightly up. So, what that means is these are both facing the same face of the ring, which would be the top, so these are actually cis. Isn't that interesting? So, I don't look at equatorial or axial. I just look at what direction it is going.
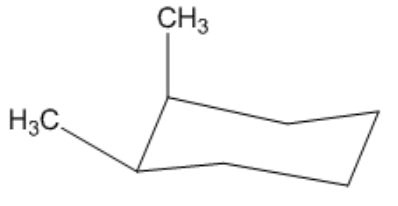
Is the following cyclohexane cis or trans?
Video transcript
Now notice these are both axial. I have axial and axial. Does that matter? No, it doesn't. These are still going to be trans. Okay? The reason they're trans is because I have one facing the top face of the ring, one facing the bottom face of the ring, so they're facing opposite directions, so that would be trans. Alright? Does that make sense to you guys? That's how easy it is. It doesn't have to get any more complicated than that. All right? So let's move on to our next topic.
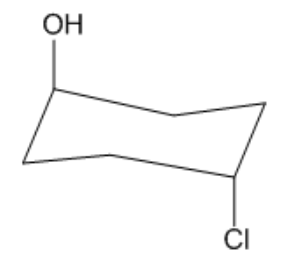
Not too bad, right? It's as easy as it sounds! Let's move on.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Name the following compounds. Remember that two up bonds are cis; two down bonds are cis; one up bond and one ...
- Given the following chair conformations, draw each in its planar form as if you were viewing it from above. (...
- Given the following chair conformations, draw each in its planar form as if you were viewing it from above. (...
- Is each of the following a cis isomer or a trans isomer? a. b. c.
- Given the following chair conformations, draw each in its planar form as if you were viewing it from above. (...
- Name the following compounds. Remember that two up bonds are cis; two down bonds are cis; one up bond and one ...
- Use your results from [PROBLEM 3-27] to complete the following table . Each entry shows the positions of two g...
- The cyclohexane chair shown in [FIGURE 3-22] <IMAGE> has the headrest to the right and the footrest to t...
- Draw two different chair conformations for each of the following molecules. Make sure that your drawings clear...
- Which of the following represents a cis isomer?<IMAGE>
- Name the following cycloalkanes using the IUPAC system of nomenclature.<IMAGE>

