A functional group that we definitely need to know how to name is alcohols. And they're really not so hard, so let's just jump straight into it. So a word that we use to describe a molecule that has more than 1 OH or more than 1 hydroxyl on the carbon chain is a glycol. Glycol is a very unspecific word because it just refers to any molecule that has more than 1 hydroxyl on it. So instead of using the term glycol, a lot of times we're going to use a more specific term. In fact, we're going to use prefixes to indicate exactly how many OHs are on that chain. So as you can imagine, we're just going to use the same prefixes that we use for all IUPAC nomenclature. If you have an alcohol with 2 hydroxyls, that's going to be called a diol. Okay? And if you have an alcohol with 3 hydroxyls, you can imagine that's going to be called a triol and that would keep going to tetra, etcetera. Okay? Now keep in mind that something that's unique about alcohol groups is that you always give the most priority or the highest priority in terms of the way that you number the chain or the way that you number the ring to the OH group. Okay? So there's actually this phrase that we use in Organic Chemistry 1 that just says alcohol beats all. Okay? And that just means going to be all the other functional groups that you're really exposed to in Organic Chemistry 1. If you have a double bond, triple bond, alkyl halide, anything else present, you're going to give your priority in terms of prioritizing numbering to the alcohol. Okay? So let's just go ahead and jump into these examples. I want you guys to try to solve it on your own and then I'll go ahead and jump in and give you guys the answer.
Alcohol Nomenclature - Online Tutor, Practice Problems & Exam Prep
We’ve learned how to name simple alcohols before, but now we are moving to polyols.
Note: I am moving away from the term glycol, and using polyols instead, since the term glycol is not specific enough.
How to name polyols.
Video transcript
In Summary:
Polyols with two hydroxyls are called diols, and polyols with three hydroxyls are called triols.
- Always give most priority to the –OH group.
Provide the correct common and IUPAC name.
Video transcript
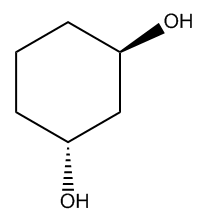
All right. So let's go ahead and start off with the root name. The root for this compound would be cyclohexane. So let's go ahead and write that down. Great. So we've got cyclohexane, but obviously, I have some substituents coming off of this. In fact, these are functional groups. I have 2 alcohols, so I can imagine that my modifier is going to be diol. So I'm just going to put here mod is diol. So I could put these two words together. And I know that this is going to be a cyclohexanediol. Okay. So I just put the modifier at the very end of the root. Now, we need locations, right? Because I don't know exactly where these alcohols are. What we would do is we would just give top priority to one of the alcohols. It doesn't matter which one you choose since they're both the same thing. But we do know that if one had been an alcohol and the other one was, for example, a halogen, I would give my highest priority to the alcohol, right? Because alcohol beats all. So let's just pick this as my one. That means that my other alcohol is going to be at the 3. So this is going to be a 1,3-cyclohexanediol. So are we done? We are so close, but there's one more thing we have to add and that's going to be the relationship of the OHs to each other in terms of where they are in space or their stereochemistry. So remember that if you have 2 groups on the same side of the ring or different sides of the ring, they get different names, right? So if they're on different sides of the ring, that would be considered trans. This would be the trans stereo isomer of 1,3-cyclohexanediol. All right? So not so bad. Let's go ahead and try to name this other example. Go ahead and try to do it on your own, and then I'll give you guys the answer.
Note: The molecule should be named trans-1,3-cyclohexanediol.
There are other possible name variations that are also acceptable. Here are just a few:(R,R)-cyclohexane-1,3-diol, (1R, 3R)-1,3-cyclohexanediol
Provide the correct common and IUPAC name.

Do you want more practice?
More setsHere’s what students ask on this topic:
Your Organic Chemistry tutors
- Predict which member of each pair has the higher boiling point, and explain the reasons for your predictions. ...
- Predict which member of each pair will be more soluble in water. Explain the reasons for your answers. (a) hex...
- Draw the structures of the following compounds. (Includes both new and old names.) (d) 3-cyclopentylhexan-3-o...
- Give systematic (IUPAC) names for the following diols and phenols. (a) (b)
- Predict which member of each pair will be more acidic. Explain your answers. (d) 2,2-dichloropropan-1-ol or 2,...
- Predict which member of each pair will be more acidic. Explain your answers. (c) 2-chloroethanol or 2,2-dichlo...
- Predict which member of each pair is more acidic, and explain the reasons for your predictions. (b) cyclohexan...
- Predict which member of each pair is more acidic, and explain the reasons for your predictions. (a) cyclopenta...
- Which of the following bases would favorably deprotonate a hydroxyl group? (b) NaOH
- Which of the following bases would favorably deprotonate a hydroxyl group? (c) NaCN
- Which of the following bases would favorably deprotonate a hydroxyl group? (e)
- (•) The following molecules were named incorrectly according to IUPAC nomenclature. Give the correct name of t...
- (••) Using IUPAC rules, name the following molecules.(d) <IMAGE>
- (••) Draw the correct structure from the following IUPAC names:(a) (4R,2Z)-4-methylhex-2-en-1-ol
- (••) Draw the correct structure from the following IUPAC names:(c) (1S,4R)-4-bromocyclohex-2-en-1-ol
- Draw a condensed structure for each of the following:g. 1-bromo-1-pentyneh. 5-methyl-2-cyclohexenol
- Give a systematic (IUPAC) name for each alcohol. Classify each as primary, secondary, or tertiary.(f) <IMAG...
- Give a systematic (IUPAC) name for each diol(a) CH3CH(OH)(CH2)4CH(OH)C(CH3)3(b) HO-(CH2)8-OH(c) <IMAGE>
- Predict which member of each group is most soluble in water, and explain the reasons for your predictions.(a) ...
- Give a systematic (IUPAC) name for each diol(d) <IMAGE>(e) <IMAGE>
- Which of the following bases would favorably deprotonate a hydroxyl group?(d) Et₃N
- Which of the following bases would favorably deprotonate a hydroxyl group?(a) <IMAGE>
- Propose mechanisms to show the interchange of protons between ethanol molecules under (a) acid catalysis. (...

