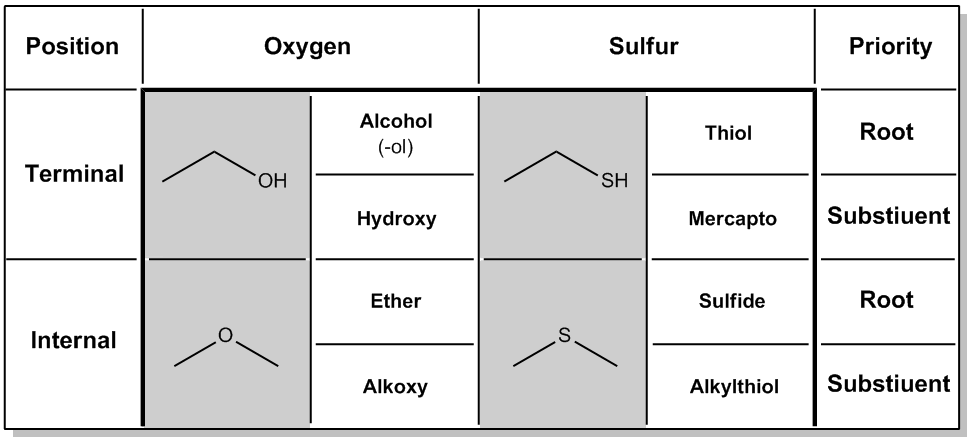
Now let's talk about the nomenclature of sulfur-containing compounds. So it turns out that sulfur-containing compounds are going to be named very similarly to oxygen compounds because of their placement on the periodic table. If you remember, oxygen is on the second row and sulfur is right below it on the third row. What that means is that many of the compounds that you've seen made with oxygens, for example, alcohols and ethers, can also be made with sulfur. But obviously, they're going to have different names. So what I've done here is I've made this nice chart that seems kind of confusing at first, but it really lists out every single possibility of a sulfur-containing compound that you could have. It turns out that there are two main functional groups that we've learned to name for oxygen. And that would be alcohols where you have a terminal OH or a terminal oxygen. Notice that it's at the end of the carbon chain. And we also have, well, if you move that oxygen inside the carbon chain, it becomes an internal oxygen or what we call an ether. So hopefully, these two names should be familiar to you guys at this point because these are oxygen-containing compounds. We usually discuss these a lot before we talk about sulfur. But it turns out that sometimes these substituents may not always get or these oxygens may not always get the highest priority. Sometimes, oxygens are actually named as substituents. And what we found is that when alcohol is named as a substituent, that means it actually has a higher priority group other than the alcohol present. Now just so you know, side note, you haven't seen a lot of these yet in this course. Okay? But let me just give you an example. A carboxylic acid would get a higher priority than an alcohol. We're going to do more of that in orgo 2 where we name an alcohol and a carboxylic acid on the same chain. For right now, I told you guys alcohol beats all because it almost always does in orgo 1. But later on in orgo 2, we will find exceptions to that. So anyway, let's say that the alcohol is a substituent. Instead of calling it alcohol, we call it hydroxy. And that makes sense. So we'd say it's a 1-hydroxy, 2-hydroxy, etcetera, etcetera when we're naming it as a substituent. In the same way, ether can also be named as a substituent. If you recall, the way that we name ethers in the IUPAC way is to name a long carbon chain and then to name the alkoxy substituent or the ether part as an alkoxy substituent. So now we've just covered everything about oxygen. Now what we're going to do is we're going to shift over here to sulfur and I'm going to show you guys how it's really the same thing. So it turns out that sulfur can also have two possibilities. It can form on the terminal, so it means it's at the very end. It only has one H or it can form on the internal part of a chain, meaning that it has R groups on both sides. So what you can see is that the sulfur at the top is a lot like an alcohol. The sulfur at the bottom is a lot like an ether. But like I said, these are not going to get the same names because then that would be very confusing. They're going to get different names. So when a sulfur is on the terminal end and it's the root, just like alcohol is the root of the name, then we're going to name it a thiol. Okay? So you can think of alcohol and thiol go together. Okay. Now how about if the sulfur is there, but I have another high priority group present, for example, like a carboxylic acid over here. Let's say that I had a carboxylic acid. So what that means is that now my SH is not getting top priority. That means it's being named as a substituent. Well, in that case, instead of calling it thiol, we're going to name it mercapto. Now I know that sounds really weird. That's a word that actually comes from the Latin and they just stuck with it. Okay. "Mercapto" means sulfur in Latin. But all you need to think is that mercapto is similar to hydroxy. If there's a situation where I used hydroxy as a substituent, I would also use mercapto as the name of the substituent. You're only going to use mercapto if there's a higher priority group present on the chain. Now finally, let's go to the ether analog. And for an ether of a sulfur, a sulfur ether, what we're going to name it is as a sulfide. Okay, so just think that ether is ROR, sulfide is RSR. Okay? I'm just going to even write that right here. RSR. Just so you guys can see that it's very similar to ether. But sometimes we don't name it as the common system. Remember that in the common system, we just name it as ether or sulfide. But sometimes we're going to use the IUPAC system. And in the IUPAC system, we would name it as an alkylthiol, which is very similar to alkoxy, just instead of oxy, it's thiol. Okay? So I know that was a lot to walk through, but hopefully by thinking of sulfur in the context of oxygen, what you already know from oxygen, that's going to make it easier for you to get a grasp on it. The only thing is there's just some funky words we have to remember. So now we have some practice problems that I want you to go ahead and do from this chart. Try to do it try to get the exact name for this first one. Try to do the whole thing and then I'll go ahead and give you the answer. One more thing. Just keep in mind that alcohol actually has a higher priority than thiols, so that should be giving you an idea of which one to name as the root. So go for it.
Naming Thiols - Online Tutor, Practice Problems & Exam Prep
Sulfur is right below oxygen on the periodic table. So that means it can form analogs of common oxygen-containing functional groups.
How to name sulfur-containing compounds.
Video transcript
Note: Alcohol has higher priority than thiols.
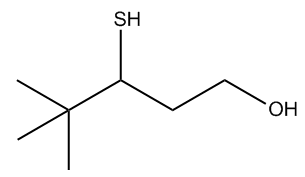
Provide the IUPAC name for the following compound.
Video transcript
Hey, guys. This video is a rerecord because the first time that I recorded this video, I actually got the question wrong. So that confused a lot of you and now we're going to try it again, okay? So what is the name of this molecule? Well, we see that we have a thiol and an alcohol on the same chain, so we have to figure out which one has priority, right? And the priority actually goes to alcohol. So what that means is that as I name this chain, I'm going to keep in mind that the one position needs to start from the side closest to the alcohol which happens to be the carbon with the alcohol, okay? Great. So what's the longest carbon chain? Let's just go through our IUPAC rules. It would be 5 carbons right here. What are our substituents? Well, this SH is considered a substituent since it's on the main chain of an alcohol, so we know that's going to get the name mercapto. And then we have 2 methyl groups, so that's going to be a dimethyl. So let's go ahead and list out what the root name is with the modifier and then what the substituents are. So we know that the root name is going to be 1-pentanol, right, that's the root. And we know that it's 1-pentanol because I have 5 carbons and the OH is in the one position. That means that if you keep counting, this is the 3 position and then this is the 4 position. So what are our substituents? I'm just going to put subs. Well, I know that I have a 3-mercapto. And I know that I also have a 4,4-dimethyl. Now when we name the entire molecule, we're going to have to put it in alphabetical order, right? So what that means is that I need to figure out is mercapto or methyl or dimethyl before the other in the alphabet. Remember, the di does not count. So all I'm looking at is the m's here. So in terms of alphabetical order, m is in the same exact place in the alphabet, so I have to go to the next letter. Let's go to e. They're both e's. Then I have to go to the next letter. It's like a playoff system. The next letter is t versus r. So this one, you might need to start saying your ABCs. But if you do, you're going to find that T is after and R is before. So that means that mercapto comes first. So that means that this entire name is going to be 3-mercapto-4,4-dimethyl-1-pentanol. Does that make sense? Cool. So that is the answer. And again, I know it sounds silly with the ABCs thing, but I would rather you say ABCs during your class or during your exam than get something like this wrong because you forgot which one goes after t or r. Okay? So we're done with this problem. Let's move on to the next.
Provide the IUPAC name and common name for the following compound.
Video transcript
So remember that it's usually easier to name ethers in the common name system, meaning that it's just both the R groups and then ether at the end. Well, the same is going to apply to sulfides. Sulfides are typically easier to name in the common name system, so let's start with that one. I'm just going to write here 'common' and let's figure it out. What are the 2 R groups? Well, in this case, I've got an ethyl on one side and a benzene ring by itself. It's not called benzene. It's called phenyl. Remember that? So I just have to put them in alphabetical order. The common name is going to be easy. This is just ethylphenylsulfide. Cool? So that one wasn't so bad. You can see how if it would have been an O there, it just would have been ether. Same thing.
Now let's go with the IUPAC name. The IUPAC name is a little bit more complicated because remember that the IUPAC name, you have to name the bulkiest or the longest root and then you have to name the SR or the OR, whichever one it is, as the substituent. So in this case, I have two different possibilities. I have either the two-carbon chain here that's ethyl or I have the six-carbon ring on the other side, which is phenyl as a substituent, which one is going to want to be the root? I'm going to go with my benzene ring because my benzene ring is overall bigger than the two-carbon chain on the other side. So, what that means is that instead of calling this a phenyl group, I'm just going to call it benzene because that's going to be the root. So now if this is benzene, I'm just going to scratch out phenyl, it becomes benzene now because that's my root. Then, what is the substituent? Well, my substituent is usually called, we said it's called an alkylthiol group. So in this case, the alkyl is just ethyl. So the name, the IUPAC name here would be ethylthiobenzene. All one word. Okay? You might be wondering, well, John, do we have to say the location of it? Do we have to say that it's like 1-ethylthiobenzene? No, because there's only one substituent, so it's always assumed that you can start at the 1. But if you wanted to, you could say as well that it is 1-ethylthiobenzene. Okay? But typically, if you see it on your exam or in the book, you'll see that the number 1 is omitted if there's only one substituent present. We usually only use locations on rings if there's more than one substituent, for example, 2 or 3. Okay? Awesome guys. So hopefully that made sense. Let's keep moving on.

Do you want more practice?
More setsYour Organic Chemistry tutors
- Draw the structures of the following compounds. (Includes both new and old names.) (i) cyclopent-3-ene-1-thiol...
- Give IUPAC names for the following compounds (a) (b) (c)
- Provide the IUPAC name for the following molecules. (f)
- (••) Using IUPAC rules, name the following molecules. (e)
- Draw the structure that corresponds to the name provided.(e) (1S,4S)-4-isopropylcyclopent-2-enethiol
- (••) Draw the correct structure from the following IUPAC names:(e) (R)-2,2-dimethyl-1-phenylpropane-1-thiol

