Alright, guys. So now let's talk about naming epoxides. It turns out that epoxides are just cyclic ethers. That's basically the definition. And some types of cyclic ethers, remember that an ether is R-O-R, are going to be named as their own functional group due to increased reactivity. Okay? And the specific ones that we usually name as their own functional group are 3-membered ethers. Okay? Because there happens to be a lot of strain in those rings. They're out of their normal bonding preferences or their normal angle preferences. So what that means is that they're very reactive and it's very easy to open them up. And what we call these 3-membered cyclic ethers is there are actually 2 common names for them. We call them epoxides. So go ahead and write that down. Okay? They're also called, in some textbooks, some professors like to use the word oxirane. Okay? Oxirane. These are really synonyms for each other. Okay? An epoxide and an oxirane are the same exact thing. It's just a 3-membered cyclic ether.
Naming Epoxides - Online Tutor, Practice Problems & Exam Prep
There are 3 distinct ways to name cyclic ethers. But before we get into them, let’s first specifically define what an epoxide is.
Defining what an epoxide (oxirane) is.
Video transcript
Cycloalkane Convention
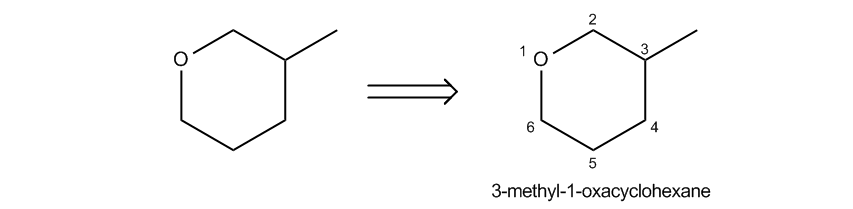
Name the ring as a cycloalkane, adding the prefix oxa- and location if necessary.
How to name cyclic ethers using the cycloalkane convention.
Video transcript
Cool so far, right? Now, the challenge becomes how do we name these guys because sometimes, first of all, they're not always three-membered rings. And second of all, there are a lot of substituents. So it turns out that there are 3 different common ways to name epoxides and I'm going to go over all of them right now. Let's start off with what we call the cycloalkane convention. In this type of naming system, what we do is we name the entire ring as if it was an alkane first. Okay? So as you can see here, I have a 6-membered ring. But how many of those atoms are actually carbons? Only 5 of them are. I've got 1, 2, 3, 4, 5. Now you might be wondering why I started the one there. I didn't need to. I'm just using that. I mean maybe I did, but I'm just using that as an example right now just to count carbons. Okay? So I have 5 carbons, but what I'm telling you is that we should actually name it as a cycloalkane, not by the number of carbons. So what that means is that usually when we're naming an alkane, we would say there are 5 carbons, so this would be cyclopentane. But it's not. We're going to call this actually cyclohexane because we go by the shape. What we're worried about here is the shape of the molecule, not how many carbons it has in it. So this would be a cyclohexane first of all as our root. Now the difference is if we have oxygens inside of a ring, which is by definition a cyclic ether, right? Then we're going to add the prefix oxa. And what oxa is going to tell us is that there is one member of this ring that is an oxygen. Okay? So if I call it oxacyclohexane, what I'm saying is that I have a 6-membered ring where one of the atoms is an oxygen, not a carbon. Okay? And then obviously location if necessary. So let's go ahead and just talk about this for a second. The root is going to be the oxacyclohexane. I have that written here. Now we just have to talk about locations. How do we know where to put those guys? Well, it turns out that the oxygen is always going to get your one spot. So when I put the one here, that didn't really count. That wasn't true numbering. The way that I should really number it is starting from the oxygen because that's the highest priority atom inside the ring. And then obviously, I should number to give the lowest overall number or to go to the next highest priority, Okay? So this would be 3-methyl-one oxacyclohexane. Cool so far? Just so you guys know, this also applies to rings that have more than 1 oxygen. If I had 2 oxygens, that would be what was called a dioxa. Okay? Just putting that out there, you could use prefixes as well. Okay? So now let's go ahead and talk about another naming system. This is going to be commonly used for non-three-membered rings. So if not three-membered as you can see, I was dealing with a 6-membered one here. This is usually the one we use. Okay? So if it's 4-membered, 5-membered, 6-membered, etcetera, you would use this naming system. Now, if it is a 3-membered ring, we could still use it but this is not going to be the most common way to name it. Okay?
Epoxy Convention
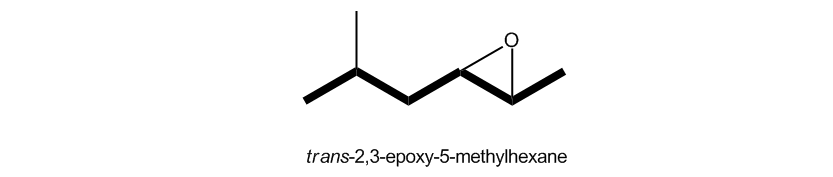
Name as a typical alkane, and then include epoxy as a di-located substituent.
How to name epoxides using the epoxy convention.
Video transcript
If we are dealing with a 3-membered ring, there are much more common ways. One is the epoxy convention. So what the epoxy convention basically says is this: We have a substituent named an epoxy group. Okay? And we're just going to name our longest carbon chain as normal and then label the 3-membered ring as just the substituent coming off of that chain. And obviously, give it the lowest number. One other thing about this that's interesting is that you actually have to name the locations of both of the atoms that the three-membered ring is attached to. So as you can see here, my epoxide is going to get priority over the methyl, so I would choose this to be my first carbon over here. That means that my epoxy group, or my epoxy substituent, is across the 2 and the 3. Therefore, I'm actually going to call this a 2,3-epoxy substituent because I'm basically saying that I have a bond O across those two carbons. Then the rest of it, we're just going to name like always. So this would be 2,3-epoxy-5-methylhexane. Not so bad, right? Okay. So it's just something to consider that you could also use the epoxy convention. It's perfectly legit.
Oxide Convention
Name as an imaginary alkene, then follow with the word oxide.
How to name epoxides using the oxide convention.
Video transcript
Now there's on top of that, there's even one more way to name epoxides and that this one actually comes from even further back in the history. This one is actually like a reaction. Okay? What they're basically saying is name it as an alkene. So pretend that the epoxy wasn't even there, replace it with an alkene. Okay? Name it as the alkene. Entire name. And then at the end, just add the word oxide. Okay? Now how does that make sense? The reason that makes sense is because what we're saying is that we're basically assuming that we start off with a double bond and then we did an epoxidation to put an epoxide group on that double bond. Now, you might not know how to do that yet and that's fine. We're going to actually learn that pretty soon. But I'm just saying that this is almost coming from the reactivity side of things saying, well, I could start from a double bond and if I do an epoxidation, I could get an epoxide, so then it would call it an oxide of that double bond. So in this case, I would call this this would be hexene because I've got a 6 membered chain. Notice that my double bond would be across the 2 and the 3, but the way that I name double bonds is different from the way that I name epoxides. I actually don't say that this is a 2-3 alkene. I would just start where at the lowest number. So in this case, this is actually going to be what we call a 2-hexene. So don't get them confused. This would never be called a 2-3 hexene. You only do that for the epoxy substituent. So we know you have a 2-hexene. Now we need a substituent, the 5-methyl. Right? On the 5, so this would be 5-methyl-2-hexene oxide. Now just so you know, if your professor requires stereochemistry, if your professor is asking about stereochemistry, remember that's just like cis and trans and stuff, then you would have to provide it here. Okay?
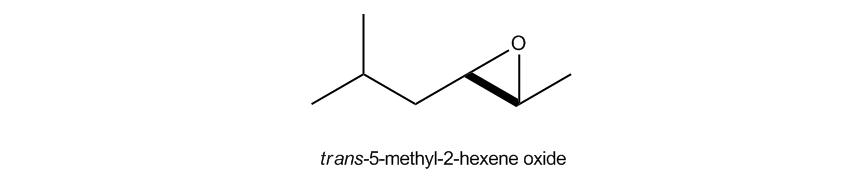
Note: I forgot to address in the videos that both of these would be trans due to the alkyl groups facing opposite sides of the ring.
Which of the following is the correct name of the following compound:

Which of the following is the correct name of the following compound:

Do you want more practice?
More setsYour Organic Chemistry tutors
- Draw structures for the following: d. 2,3-epoxy-2-methylpentane
- Draw structures for the following: c. 2,2,3,3-tetramethyloxirane
- Write structural formulas for the following compounds. (g) cis-2,3-epoxyhexane (h) (2R,3S)-2-methoxypentan-3-o...
- Draw structures for the following:a. 2-propyloxiraneb. cyclohexene oxide
- Give two names for each of the following:a. <IMAGE>b. <IMAGE>
- Name the following: c. d.
- Give common names for the following compounds.(g) <IMAGE>
- Give IUPAC names for the following compounds.(g) <IMAGE>(h) <IMAGE>(i) <IMAGE>
- What is the major product obtained from the reaction of 2-ethyloxirane with each of the following reagents?a. ...

