In this section, we're going to talk a lot about alcohols. So it's important for us to take a minute to remember all the different ways that we can make alcohols from prior chapters. So let's go ahead and get started. It turns out that there are 4 different ways that we've already learned to make alcohols. And I'm going to go ahead and remind you of what those are now. So let's say that I have a double bond and I'm trying to add a hydroxyl group to it. And I want to add it specifically in this position, what addition reaction could I use to put a hydroxyl right on that secondary position? The answer is that there's only one reaction that could yield this kind of alcohol and that would be oxymercuration or what we just called oxymerc. Why is that? Because if you remember, oxymercuration is an addition reaction that adds Markovnikov alcohol, which is good because you can tell that that is the Markovnikov or more substituted location. But there's another thing about oxymerc, which is that there's no carbocation in the intermediate. Okay? And remember that carbocations have a tendency to do what? They shift. So in this case, this is an example of where I can add Markovnikov alcohol without worrying about a rearrangement. So now all we have to do is fill in the reagents for oxymerc. Does anyone remember them? Well, basically what we would have is some kind of mercury molecule, so it would be HgOAC2. And then what we would put with that is water. Why is that? Because we want to show specifically that we're using water to add the alcohol. On the bottom of the arrow, then we did the reduction step. Remember that the full name of this reaction is oxymercuration reduction. The reduction is completed by NaBH4, which later on we will learn as a reducing agent and a base, so NaOH. Okay? Those are the reagents. I know it looks like a lot, but we have been over this before and this is one of the ways that you can add alcohol to a double bond. All right? So we've been over one of the 4 ways, but let's keep going because it turns out that there's a lot more that we can do to a double bond.
Alcohol Synthesis - Online Tutor, Practice Problems & Exam Prep
Up to this point in the course, we’ve learned 4 methods to make alcohols. This will be a brief review of those methods.
Forming alcohols through Oxymercuration-Reduction.
Video transcript

Forming alcohols through Hydroboration-Oxidation.
Video transcript
So what if we take that same double bond, but we want to add the alcohol here instead? Notice that in this molecule, I have the alcohol on the least substituted position. Does anyone remember what kind of reaction that would be? Well, it's going to be another addition reaction but has a completely different name. This is hydroboration. Remember hydroboration is really unique because it's going to be the only anti Markovnikov way we have to add alcohol to the least substituted position. Okay? So let's just go ahead and remember what the reagents were. Does anyone remember? It was really hydroboration means that you need boron in there. And it was really going to be any boron source, most commonly BH3, but your professor, your textbook may use a different source of boron. There are all kinds of different sources that we can use. And BH3 and many times that will be complexed to THF. But sometimes you won't see that step, but sometimes THF is a solvent, so it's not absolutely required that you write that. But sometimes you'll see it. And that's it for the top. The top is just adding boron. The bottom is the oxidation step because remember that it's hydroboration oxidation. Let's fill those reagents in. That was hydrogen peroxide and once again a base. Okay? So those are the reagents that could generate an anti Markovnikov alcohol from a double bond.
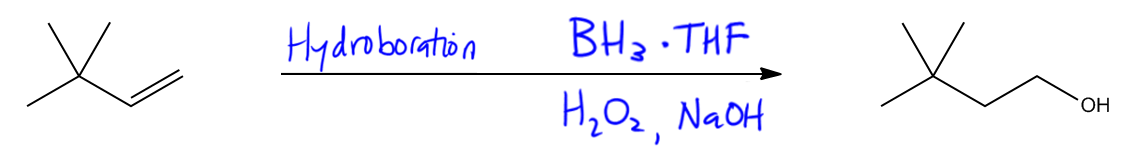
Forming alcohols through Acid-Catalyzed Hydration.
Video transcript
Cool. So we've gone over 2 ways. Now let's go on to the next one. Notice that let's say I start off with that double bond, but now what I end up getting is an alcohol not in the secondary position, but in the tertiary position. That's kind of odd. Notice that the double bond used to be here, but now my alcohol isn't even attached where the double bond was before. Okay? Any clue what this might be? This is also going to be a Markovnikov addition, but it's a Markovnikov addition that also has a carbocation in the intermediate in the reaction. So what this means is that this is going to have to be an acid-catalyzed hydration. Sorry. Acid-catalyzed hydration. There we go. And the reagents for this one were much easier. It was really just any strong acid. So I'm just going to put here over water. But most commonly, the acid that we would use is sulfuric acid. So most commonly what you're going to see is, like H2O over H2SO4 or the other way around too. Both are fine. Okay? So that's usually what you would see. And remember what that's going to do is it's going to add water, but it's going to add it to the most stable location, which is the one that is created. The tertiary position is created after it shifts. So those are the 3 different ways that we have to make alcohol from an addition reaction. And this is just meant to be an overview of the reactions that we've already learned. If you need to brush up on your addition reactions, I would recommend to go to the addition reactions chapter and I always teach all these 3 in a row as well, so you'll be able to really learn the full mechanisms and catch up on some of the stuff that you might be rusty on.

Forming alcohols through SN2 reactions.

Do you want more practice?
More setsYour Organic Chemistry tutors
- Show how you would synthesize the following alcohol from appropriate alkene. (a)
- Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the fol...
- Predict the product of each of the following alcohol synthesis reactions. (b)
- Show a mechanism for each of the alcohol synthesis reactions in Table 13.8.<IMAGE>
- a. Show the reagents required to form the primary alcohol in each of the following reactions.
- Suggest a reagent and a reactant that could be combined to make each of the following alcohols.(c) <IMAGE&g...
- Suggest a reagent and a reactant that could be combined to make each of the following alcohols.(d) <IMAGE&g...
- Predict the product of each of the following alcohol synthesis reactions.(c) <IMAGE>
- (•••) Starting with formaldehyde and the alkyl halides shown as the only sources of carbon, provide a synthesi...
- Show how you would synthesize the following alcohol from appropriate alkene.(d) <IMAGE>
- Show how you would synthesize the following: (c) cyclohexylmethanol from an alkyl halide using an SN2 reactio...

