So now we're going to switch gears and move away from talking about strict addition reactions to talking about oxidation reactions. It turns out that double bonds can not only be added to but they can also be oxidized. What that means is that oxygens can be placed directly on them. So, what we're going to talk about is some different ways to do that and what I want to talk about right now is ozonolysis. Ozonolysis would be categorized as a reaction that is a form of weak oxidative cleavage. Basically, cleavage just means to cut something. This entire time we're going to be using our visual scissors to cut things in different pieces. There is a mechanism for ozonolysis; it's very long. If you need to know it for your professor, believe me, I'll teach it to you. But that's not going to be on this page. On this page, I just want to give you a general overview of what ozonolysis does. Ozonolysis basically slices double bonds in half and winds up making a combination of ketones, aldehydes, and formaldehyde. Why does it make each of those things? I'll explain. But think about it this way: imagine that you have a very long carbon chain with one double bond in the middle and you cut it into 2 − how many chains would you expect to have? 2. That's pretty easy. How about if I have a ring and I have a double bond in one part of it and then I snip it right there? What would I expect to get at the end... Two rings? Two chains? No, I would just expect one chain because I have a ring and I cut it in one place, so now I have one chain. These are just simple geometry questions that actually get students confused with ozonolysis. That's exactly the way this works. So here, you can see I have a 7 carbon chain with 2 places to cut. I could use scissors here and I could use scissors here. Please, the scissors reference is just to help you visualize what's going on. What winds up happening is that if we have a 7 carbon chain with 2 cuts, I'm going to wind up getting 3 pieces. I'll get one piece at the end, a piece in the middle, and a piece on the other end. Does that make sense? The number of carbons at each end is going to be the number of carbons in the product. Here along this cut, I'm splitting it off with 1 carbon at the end. Here between the two cuts, I've got 4 carbons in the middle. And then here at the top, I've got 2 carbons being chopped off. So, I'm going to expect 3 different products: a 1 carbon product, a 4 carbon product, and a 2 carbon product. Now let's look at the reagents really quick. The reagents are actually really easy for ozonolysis because it's always just going to be ozone. O3 is ozone. So anytime you see that, you know this is ozonolysis. Sometimes different professors use different reagents as the reductive workup. Some will use zin
10. Addition Reactions
Ozonolysis
10. Addition Reactions
Ozonolysis - Online Tutor, Practice Problems & Exam Prep
Ozonolysis is a form of oxidative cleavage that breaks through alkenes and replaces them with carbonyls on both ends. Just imagine the ozone functioning like tiny atomic scissors, cutting each alkene right in half!
1
concept
General properties of ozonolysis.
Video duration:
6mPlay a video:
Video transcript
The products of ozonolysis are a mixture of ketones, aldehydes and formaldehyde. You get these with:
- Ends of alkenes with –R groups on both sides = Ketones
- Ends of alkenes with 1 –H = Aldehydes
- Ends of alkenes with 2 –Hs (yielding single carbon fragments) = Formaldehyde
General Reaction:

2
Problem
ProblemPredict the products of the following reaction.
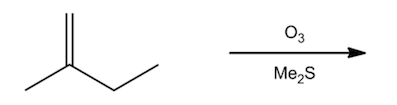
A

B

C

D

Do you want more practice?
More setsYour Organic Chemistry tutors
Additional resources for Ozonolysis
PRACTICE PROBLEMS AND ACTIVITIES (68)
- The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfid...
- For each compound, show the products obtained from ozonolysis, followed by treatment with dimethyl sulfide. b....
- For each compound, show the products obtained from ozonolysis, followed by treatment with dimethyl sulfide. a....
- What products are formed when the following compounds react with ozone and then with dimethyl sulfide? b.
- What products are formed when the following compounds react with ozone and then with dimethyl sulfide? a.
- What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide? b.
- For each compound, show the products obtained from ozonolysis, followed by treatment with dimethyl sulfide. d....
- For each compound, show the products obtained from ozonolysis, followed by treatment with dimethyl sulfide. c....
- What is the major product of each of the following reactions? f.
- "Ozonolysis of an alkene, followed by treatment with dimethyl sulfide, forms the following product(s). Identif...
- What hydrocarbon forms the following products after reaction first with ozone and then with dimethyl sulfide?
- Give structures of the alkenes that would give the following products upon ozonolysis–reduction. b. and
- Give structures of the alkenes that would give the following products upon ozonolysis–reduction. a.
- Give the products expected when the following compounds are ozonized and reduced. c. d.
- Give the products expected when the following compounds are ozonized and reduced. a. b.
- Limonene is one of the compounds that give lemons their tangy odor. Show the structures of the products expect...
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- Predict the major products of the following reactions. a. (E)-3-methyloct-3-ene + ozone, then (CH3)2S Proble...
- When compound Z is treated with ozone, followed by dimethyl sulfide and washing with water, the products are...
- Draw the product(s) you'd expect when each of these alkenes is treated first with O₃, then with Ch₃SCH₃ (a)
- Formation of the molozonide can be expected to proceed stereospecifically. Why is this the case? Show the two ...
- In Solved Assessment 9.30(b), we came up with an alkene that under the conditions of ozonolysis would produce ...
- Suggest a synthesis of the following aldehydes or ketones using the ozonolysis reaction of an alkene. (a)
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- Propose a synthesis of the carbonyl(s) using the (i) ozonolysis pathways. (a)
- Propose a synthesis of the carbonyl(s) using the (i) ozonolysis pathways. (b)
- Predict the reagents or reactant(s) necessary to complete the following syntheses. (b)
- Draw the product(s) you'd expect when each of these alkenes is treated first with O₃, then with Ch₃SCH₃(d) <...
- (••) Predict the product of ozonolysis of the triglyceride shown.<IMAGE>
- Predict the product of the following aldehyde/ketone syntheses.(a) <IMAGE>
- (•••) Ozonolysis of an unknown alkene A gives the products shown. Predict the product that results from hydrog...
- (••) Suggest reagents to carry out the following transformation. [One of the answers should be from Chapter 9....
- Draw the product(s) you'd expect when each of these alkenes is treated first with O₃, then with Ch₃SCH₃ (b) &l...
- Suggest a synthesis of the following aldehydes or ketones using the ozonolysis reaction of an alkene.(b) <I...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- Draw the product(s) you'd expect when each of these alkenes is treated first with O₃, then with Ch₃SCH₃(c) <...
- b. What alkenes would give only an aldehyde with four carbons as a product of oxidative cleavage
- What are the products of the following reactions? Indicate whether each reaction is an oxidation or a reductio...
- What are the products of the following reactions? Indicate whether each reaction is an oxidation or a reductio...
- For each compound, show the products obtained from ozonolysis, followed by treatment with dimethyl sulfide.e. ...
- What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?a. <IMAG...
- What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?c. <IMAG...
- b. The following products were obtained from the oxidative cleavage of a diene. What is the structure of the d...
- What products are formed when the following compounds react with ozone and then with dimethyl sulfide?c. <I...
- The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfid...
- The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfid...
- One of the constituents of turpentine is α-pinene, formula C10H6. The following scheme (called a “road map”) g...
- Problem 8-54 describes a new method to perform ozonolysis reactions that used pyridine (py) to generate the fi...
- Problem 8-54 describes a new method to perform ozonolysis reactions that used pyridine (py) to generate the fi...
- Deduce the structure of each compound from the information given. All unknowns in this problem have molecular ...
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- Professor Patrick Dussault (University of Nebraska at Lincoln) has developed an alternative to the standard tw...
- Give structures of the alkenes that would give the following products upon ozonolysis–reduction.c. <IMAGE&g...
- Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds...
- Show how you would synthesize each compound using methylenecyclohexane as your starting material. <IMAGE>...
- Ozonolysis can be applied selectively to different types of carbon–carbon double bonds. The compound shown bel...
- Professor Patrick Dussault (University of Nebraska at Lincoln) has developed an alternative to the standard tw...
- Predict the major products of the following reactions.c. <IMAGE> + O3, then (CH3)2Sd.1-ethylcycloheptene...
- What products are formed when the following compounds react with ozone and then with dimethyl sulfide?e. <I...
- Ozonolysis of an alkene, followed by treatment with dimethyl sulfide, forms the following product(s). Identify...
- What is the major product of the reaction of 2-methyl-2-butene with each of the following reagents?d. O3, −78 ...
- What aspect of the structure of the alkene does ozonolysis not tell you?
- What reagents are needed to carry out the following syntheses?
- In contact with a platinum catalyst, an unknown alkene reacts with three equivalents of hydrogen gas to give 1...

