Now I want to talk about a reaction that's so similar to halogenation that a lot of people get it confused with halogenation. It's just a little bit different because in this case, we're going to be adding a halogen in the presence of water to a double bond. And this reaction is going to be called a halohydrin formation. Okay? So let's go ahead and get started. Basically, the general reaction is that I still have my double bond. I still have my diatomic halogen. The only difference is that I'm running this reaction in the presence of water instead of an inert solvent. I actually have water present. What that means is that the water is going to wind up interacting and my end product is going to give me an alcohol on one side and a halogen on the other. This molecule right here is called, as a functional group, it's called a halohydrin. Okay? So let's go ahead and talk about the actual mechanism. The intermediate is once again going to be a bridged ion, just like before. The stereochemistry is going to be anti, just like before because anytime you're opening up a 3-membered ring, you're going to wind up getting anti products and my product is going to be a halohydrin. Okay? Are there going to be rearrangements? Nope. Okay. So no rearrangements because there's no carbocation. And then finally, this actually will have Markovnikov regiochemistry because now I am adding 2 different things. So now I do have to be aware of which one goes on the Markovnikov side and which one goes on the anti-Markovnikov side. But overall, we would say this is a Markovnikov reaction because it's driven by the stability of the intermediate. Okay?
Halohydrin - Online Tutor, Practice Problems & Exam Prep
This is an indentical mechanism to halogention, except with water as the nucleophile in the second step. Why would water prefer to react as a nucleophile over a halogen anion? Let's find out.
General properties of halohydrin formation.
Video transcript
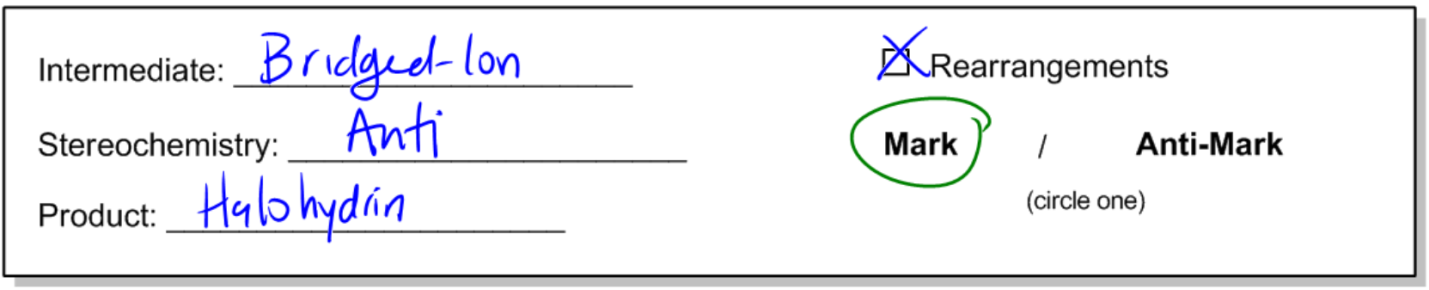
- Opening of 3-membered intermediates/molecules always results in anti-addition.
General Reaction:
Halohydrin Mechanism
Video transcript
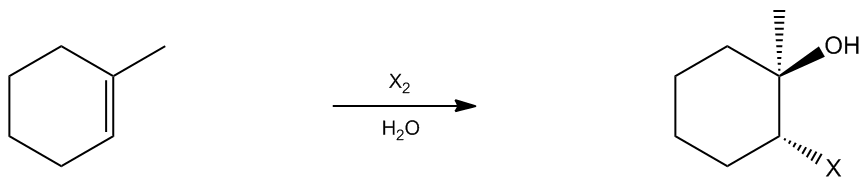
So let's just go ahead and get started. The reaction is really straightforward. What I've got here is I've got the same situation where I've got a double bond and I've got a diatomic halogen and I've also got water, let's just say. So I've also got water. Which of these is going to react with my double bond? Well, water by itself doesn't really do anything to double bonds. Now if I had water and acid, that would be different. Okay? But this is just water by itself, so you can't really react with water in a double bond. But we know that we can react a diatomic halogen. So I'm going to draw my 3 arrows once again where the X is making a bridge to the double bond and it's also kicking out one of the X's as a leaving group. What this is going to give me is a bridge ion called a halonium ion once again and now we just have to figure out what's the nucleophile that's going to do the backside attack or the nucleophilic attack of this ring. Okay? So basically, I've got 2 nucleophiles. I've got X−, just like before when we were talking about halogenation, we've got the X− just like before. Nothing has changed. But now what I also have is I've got some water lying around. Now of these 2 different species, which of them do you think is going to be the stronger nucleophile? The X− or the X− is going to be much stronger. In fact, X− is one of the best nucleophiles around. Water is kind of like because X− has a negative charge. Water is neutral. So we would expect X− to be a lot stronger. So why wouldn't I just get the X− attacking and get a halogenation reaction like before? Well, the answer is that I will. Some of that actually will happen. Some of the X− will attack and I will get halogenation. But that's not going to be what happens the majority of the time. The majority of the time, this bridged ion is so unstable that it's going to react with the first nucleophile that it encounters, even if it's not the strongest. And what if I have, once again, how about if I have a billion times more of the water than I have of the X−? So what if there's water everywhere and there's only a few X−? Okay. What's going to wind up happening? What's going to wind up happening is that even though the X− is stronger, the water is just going to have an advantage because there's a lot more of it around. Because when I planned out my reaction, I used a little bit of diatomic halogen and I put a lot of water in there. Okay? So what that means is that in the second step, even though the X− is more stable and is more nucleophilic, my water is going to wind up attacking the most substituted side. Does that make sense? Because there's just a whole lot more of it around. Now for this water, is it going to attack the more substituted or the less substituted? It's still going to attack the more substituted side because that's the one with the most positive character. And remember the water has the electrons on it. So I go ahead and I make that bond, I break that bond and what that's going to give me is a major product that is a halohydrin. So if my water adds to the front, then that means that my X is going to add to the back. Okay? And that means that if my water was in the front, then my methyl group would also be towards the back. Okay? Now is this my final product? No. I still have one more step, unfortunately, because I added water, so I need to deprotonate. Right? I need to get rid of that H. What can I use to get rid of one of those H's? I could use the X−. So I'm going to use the X− in this last step to pull off an H and give the electrons to O. So now what I'm going to get is a product that looks like this. A Markovnikov alcohol and an anti Markovnikov halogen, that's anti. Do we have chiral centers here? Yep. So we have to draw both enantiomers. The other enantiomer would be that the water attacked from the bottom, x's now at the top, and that means that my x is now at the top. And these would be a racemic mixture because I really have no clue which side it came from. Does that make sense, guys? So I hope that you guys were able to see the similarities between halogenation and the halohydrin formation. The only difference is that I've got a lot of water around, so in that second nucleophilic attack, water is going to do the attack instead of the x−. Alright? So let me know if you have any questions on that, but if not, let's go ahead and move on.
1. Electrophilic Addition

2. Nucleophilic Substitution (SN2) and Deprotonation

Predict the product of the following reaciton.





Predict the product of the following reaction.
Do you want more practice?
More setsYour Organic Chemistry tutors
- What is the major product of each of the following reactions? b.
- What is the major product of each of the following reactions? b.
- What is the major product of each of the following reactions? a.
- What is the major product of each of the following reactions? e.
- Which stereoisomer of 3-hexene forms (3S,4S)-4-bromo-3-hexanol and (3R,4R)-4-bromo-3-hexanol when it reacts wi...
- Predict the major product(s) for each reaction. Include stereochemistry where appropriate. c. cis-but-2-ene +...
- Predict the major product(s) for each reaction. Include stereochemistry where appropriate. a. 1-methylcyclohe...
- Propose a mechanism for the addition of bromine water to cyclopentene, being careful to show why the trans pro...
- Show how you would make the following compounds from a suitable cyclic alkene. d.
- Show how you would accomplish the following synthetic conversions. a. 3-methylpent-2-ene--> 2-chloro-3-met...
- b. Predict the product of formula C7H13O from the reaction of this same unsaturated alcohol with bromine. Prop...
- Suggest an alkene that could be used to make each of the following halohydrins. (a)
- Predict the product(s) of each of the following reactions, making sure to indicate the relative stereochemical...
- Provide arrow-pushing mechanisms for Assessments 9.10(b) and 9.10(c) that rationalize the regioselective and s...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) At the beginning of Chapter 9, we stated that after finishing Chapters 8 and 9, we would have the ability...
- (••) When alkenes react with bromine in water, a halohydrin is produced. When water is replaced with methanol ...
- Explain why water attacks the carbon of the bromonium ion as opposed to the bromonium ion itself in the second...
- Suggest an alkene that could be used to make each of the following halohydrins.(c) <IMAGE>
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
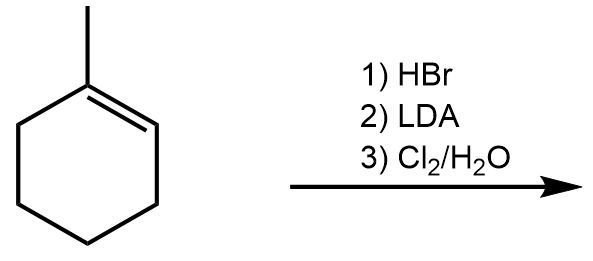
- Predict the product of the following haloalkane syntheses.(e) ↓ Cl₂₊ <IMAGE> H₂O
- Halohydrin formation is a stereospecific reaction. Identify the products of halohydrin formation of the follow...
- (•••) In light of your answer to Assessment 9.47, predict the product of the following reactions we have seen ...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (•••) Retrosynthetic analysis is the process of working backward to develop the synthesis of a new compound. I...
- What will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carried out ...
- 1-Methylcyclohexene forms two products when it reacts with bromine in methanol.a. Draw the mechanism for the f...
- Using any alkene and any other reagents, how would you prepare the following compounds?d. <IMAGE>
- What will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carried out ...
- Each of the following reactions has two nucleophiles that could add to the intermediate formed by the reaction...
- What is the major product of the reaction of 2-methyl-2-butene with each of the following reagents?j. Br2 >...
- A graduate student attempted to form the iodohydrin of the alkene shown below. Her analysis of the products sh...
- Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds...
- Show how you would synthesize each compound using methylenecyclohexane as your starting material.<IMAGE>...
- The solutions to [SOLVED PROBLEM 8-5] <IMAGE> and [SOLVED PROBLEM 8-6] <IMAGE> showed only how one...
- Predict the major product(s) for each reaction. Include stereochemistry where appropriate.e. 1-methylcyclopent...
- The solutions to [SOLVED PROBLEM 8-6] <IMAGE> showed only how one enantiomer of the product is formed. F...
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- a. Draw the product or products that will be obtained from the reaction of cis-2-butene and trans-2-butene wit...
- Show how you would accomplish the following synthetic conversions.b. chlorocyclohexane--> trans-2-chlorocyc...
- What reagents are needed to carry out the following syntheses?
- Draw the products of the following reactions, including their configurations: