So now we're moving on to yet another addition reaction, and this addition reaction is going to be one that adds two alcohols to the same double bond, and it's going to do it all in one step. And this reaction is called synvicinol dihydroxylation. Okay? So how does this work? Basically, we have these two very special reagents. We have potassium permanganate and we have osmium tetroxide, This is KMnO4 and OSO4 that are both highly capable of adding oxygens to double bonds. Now it turns out that this mechanism, you're not going to need to know the whole thing. Okay? But what you do need to know is you need to know what the end product is going to look like. And as you can see, it's really easy. All we have is a double bond. We add one of these two reagents, it doesn't matter which one. Sometimes you're going to see catalytic NMO, that's just a catalyst that sometimes comes up, sometimes it doesn't. Don't worry too much about it. And what you're going to get at the end is what we call diols. Why? Because there are two alcohols in the same molecule. They have a vicinal relationship, which means that they're next to each other, they have a one, two relationship, and they are syn to each other or they're cis, and that means the reaction was syn. Okay? So how does this actually work? Okay? The reason this works is because potassium permanganate and osmium tetroxide both have a very, very similar structure, where it's basically one central atom surrounded with as many oxygens as possible. Now, the way that I like to think of this, maybe just to make it really kind of fun, is I kind of visualize that these look like spaceships. So this is like a flying saucer and there's a flying saucer and there's like a little like cockpit here with an alien inside. But of course he's got like four arms because aliens don't have the same amount of arms as we do. And whatever. So we've got these UFOs, and what do they do? Well, they fly down to earth and they go attacking double bonds. Alright? So this UFO is like coming, it's like swooping down on the double bond, and it decides, hey, I'm gonna leave humans a gift. I'm gonna leave this double bond a gift. And what it's gonna do is it's just gonna add an oxygen to both sides of the double bond. So they're going to take one of the oxygens here, one of the oxygens there, and add them to both sides, leaving a diol at the end. Okay? So they're going to leave two alcohols behind and they're going to take off back into space. Alright? Now if you do want to know the mechanism really quick, I am gonna show you the first step. You don't need to know the whole thing, but it's going to look like this, where if you have a double bond and you have, let's say we're using osmium tetroxide, so double bond. K. What's gonna happen is you're gonna get a cyclization reaction where my, and there's some methyl there, I'm sorry, where my double bond grabs one of the O's, these electrons go down onto the osmium. Okay? And then these electrons go and make a bond to the double bond. Okay? So what winds up happening is that we get this cyclization reaction, you wind up getting two alcohols at the end, and then your OSO4, I mean, obviously it's missing two oxygen, that thing just leaves and it goes back into space. Alright? So you might need to know the first step, but regardless you're never going to need to know the whole mechanism. Alright? At least I've never seen that and I've been teaching Ordo for a very long time at a lot of universities. Alright? So let's go ahead and do a multi-step reaction here, and what I want to do is just literally, you know, just have you guys go ahead and try to figure this out by yourself, and then I'll go ahead and jump in.
10. Addition Reactions
Dihydroxylation
10. Addition Reactions
Dihydroxylation - Online Tutor, Practice Problems & Exam Prep
In this reaction, we learn how to use certain agents to add diols to a double bond. This is also known as the 1,2-syn diols reaction.
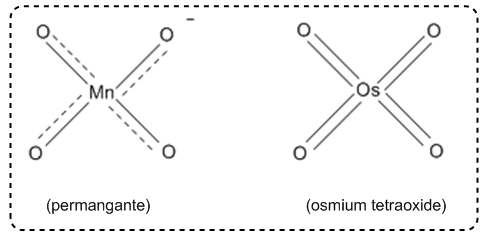
1
concept
General properties of syn vicinal dihydroxylation.
Video duration:
3mPlay a video:
Video transcript
General Reaction:
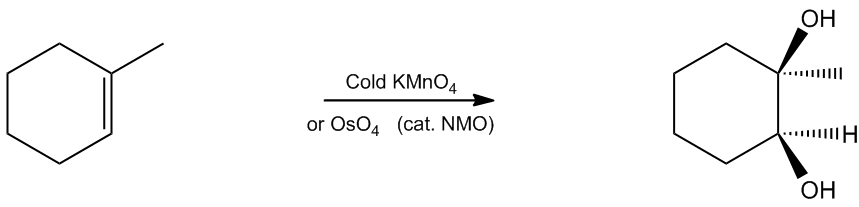
You don’t need to know this entire mechanism, but I would suggest knowing the first step:
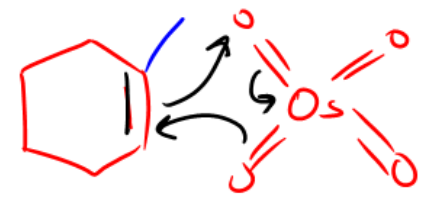
2
example
Predict the product for the following multi-step reaction.
Video duration:
4mPlay a video:

Do you want more practice?
More setsYour Organic Chemistry tutors
Additional resources for Dihydroxylation
PRACTICE PROBLEMS AND ACTIVITIES (47)
- Show how you would accomplish the following conversions. d. trans-hex-3-ene to (d,l)-hexane-3,4-diol
- Show how you would accomplish the following conversions. a. cis-hex-3-ene to meso-hexane-3,4-diol
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- Predict the major products of the following reactions, including stereochemistry. c. cis-pent-2-ene + OsO4/H2...
- Predict the major products of the following reactions, including stereochemistry. a. cyclohexene + KMnO4/H2O ...
- Show how you would make the following compounds from a suitable cyclic alkene. b.
- The two butenedioic acids are called fumaric acid (trans) and maleic acid (cis). 2,3-Dihydroxybutanedioic acid...
- Show how you would make the following compounds from a suitable cyclic alkene. a.
- Predict the major products of the following reactions, and give the structures of any intermediates. Include s...
- Predict the product(s) of each of the following reactions, making sure to indicate the relative stereochemical...
- Predict the product(s) of each of the following reactions, making sure to indicate the relative stereochemical...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) At the beginning of Chapter 9, we stated that after finishing Chapters 8 and 9, we would have the ability...
- (••) At the beginning of Chapter 9, we stated that after finishing Chapters 8 and 9, we would have the ability...
- The concertedness of the OsO₄ reaction results in both oxygens being added to the same face of the molecule (i...
- Predict the product(s) of each of the following reactions, making sure to indicate the relative stereochemical...
- When using a terminal alkene under the conditions shown here, explain why it is unnecessary to show the relati...
- Which of molecules A–D would you expect to give a positive permanganate test? That is, which would result in a...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- Draw the organic products you would expect to isolate from the following reactions (after hydrolysis). (n)
- (••) Predict the product(s) that would result when the following molecules are allowed to react under the foll...
- Suggest a reagent and a reactant that could be combined to make each of the following alcohols. (b)
- Predict the product of each of the following alcohol synthesis reactions. (d)
- Propose mechanisms for the epoxidation and ring-opening steps of the epoxidation and hydrolysis of trans-but-...
- We have studied a number of pericyclic reactions previously. Draw the mechanism of the steps shown. The sectio...
- Draw the structure of and name the enantiomeric diols that result from the cis-dihydroxylation of the alkene s...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- (••) Predict the product(s) that would result when the alkenes are allowed to react under the following condit...
- Which of molecules A–D would you expect to give a positive permanganate test? That is, which would result in a...
- How might you make the catalytic cycle in Figure 9.31 more sustainable while still using NMO as the co-oxidant...
- Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds...
- Show how you would synthesize each compound using methylenecyclohexane as your starting material. <IMAGE>...
- Predict the major products of the following reactions, including stereochemistry.e. trans-pent-2-ene + OsO4/H2...
- Limonene is one of the compounds that give lemons their tangy odor. Show the structures of the products expect...
- The two butenedioic acids are called fumaric acid (trans) and maleic acid (cis). 2,3-Dihydroxybutanedioic acid...
- Using 1,2-dimethylcyclohexene as your starting material, show how you would synthesize the following compounds...
- Predict the major products of the following reactions.f. 1-ethylcycloheptene + cold, dilute KMnO4
- The two butenedioic acids are called fumaric acid (trans) and maleic acid (cis). 2,3-Dihydroxybutanedioic acid...
- Devise a synthesis for each compound, starting with methylenecyclohexane and any other reagents you need.(c) 1...
- What stereoisomers are obtained from the reaction of each of the following alkenes with OsO4 followed by aqueo...
- (•••) Synthesize the following molecules beginning with only organic molecules containing three carbons or few...
- (•••) Retrosynthetic analysis is the process of working backward to develop the synthesis of a new compound. I...
- Show how you would accomplish the following conversions. c. trans-hex-3-ene to meso-hexane-3,4-diol
- Predict the major products of the following reactions, including stereochemistry where appropriate.(k) cyclope...

