In this video, I'm going to introduce a bunch of new addition reactions that all pretty much do the same thing. So I'm going to group them together in a type of reaction called cyclopropanation. A cyclopropanation reaction takes place when a double bond encounters either a carbene or a carbonoid. The product from this reaction is really just a cyclopropane. So we literally get the addition of an alkyl group on that double bond and all you're really getting is a methylene group or a CH2. So you might be wondering, Johnny, why would a double bond want to react with a methyl group? I mean I don't remember methyl groups being strong electrophiles. Well, that's exactly why we need a carbene or a carbonoid because methyls aren't reactive, but carbenes are. If you recall, carbenes are reactive intermediates not because of their formal charge. If you look, this is an example of a carbene right here. Does it have a formal charge? No. It has a formal charge of 0. So you might think this doesn't look reactive to me. But remember, it has a big problem. It violates the octet rule. So even though it doesn't have a formal charge, it wants to have 8 octet electrons around it. And right now it only has 6. It has that lone pair, those 2 bonds. It's missing 2 whole electrons. So carbenes are going to be extremely reactive with pretty much everything including double bonds, which is why they're going to work to make these triangle-shaped products. So what I'm going to do is I'm just going to go 1 by 1 down the list of all the reagents that can make a cyclopropanation.
Carbene - Online Tutor, Practice Problems & Exam Prep
Cyclopropanation is the general name for a series of reactions that all produce very similar products by different mechanisms. You may be responsible for one or more of these reactions on your exam, so let’s get into them.
General properties of cyclopropanation.
Video transcript
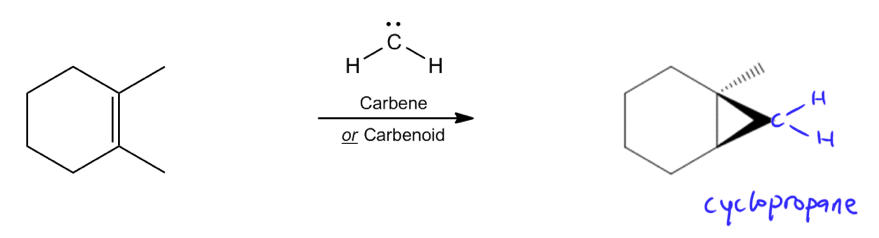
Reaction with a simple carbene.
Video transcript
Let's start at the first one. The first example would just be the easiest example possible, reacting a carbene directly with a double bond. This mechanism is going to look just like our other bridged ion mechanisms that made 3-ring intermediates. It's going to be a double bond grabbing the carbene and then the carbene grabbing back. This is going to show us that our product is cyclic. Now, whenever you add a ring to another ring, that ring has to be cis because if it was trans, you'd break the ring by having to straddle both sides of the original ring.
I'm just going to go ahead and draw this as a cis triangle or cis cyclopropane. I'm drawing it as going towards the top and then I would draw my methyl groups that were originally there going down. Now, this is not a chiral molecule, so that's my final answer. But if I had had some kind of asymmetry, then I would draw the enantiomer or just the stereo isomer that would be faced the other way.
Now, one thing to keep in mind about this, this is deceptively easy but there's one thing you need to keep in mind. These hydrogens could be swapped for any other atom that just likes to have one bond. For example, halogens, I could easily use a carbene that has let's say Cr2. If I did that, what would we need to add to this molecule? You would need to draw those bromines, guys. Here I'm just going to draw hydrogens since that's what I was using. But keep in mind that if it had been bromines or anything else, you would have to add them to the tip of the cyclopropane.

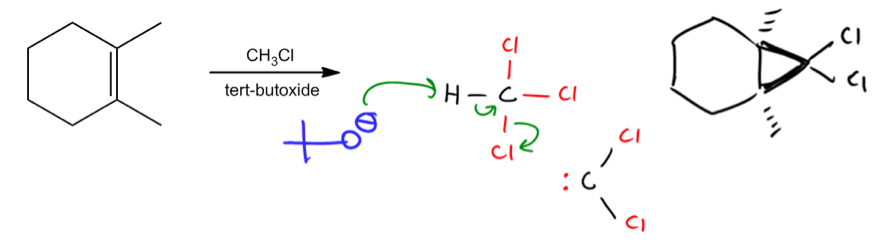
Reaction with chloroform (CHCl3) and tert-butoxide.
Video transcript
All right. So now let's move on to the second reaction that does a cyclopropanation. And I think this one's actually the trickiest one because if you look at these reagents, what you'll find is that they look strangely familiar. You have this molecule that kind of looks like an alkyl halide or a leaving group. You have this molecule that looks like a nucleophile or a base. And you might be thinking that this is in the category of substitution or elimination. You might think this is a flowchart question. Remember that flowchart that we use for those types of reactions? Well, it's not though because if you recognize that this is not a typical leaving group. Typically, leaving groups should just have 1 alkyl halide or 1 sulfony ester. But here I have 3 chlorines. That's not usual. That would not be a flowchart question. So let's go ahead and look at the generation of this carbene so that you guys can see how it works.
Basically, the way this mechanism works is that let's say you've got your carbon and you've got your hydrogen and then you've got your 3 halogens, chlorine. What happens is that this going to react in an acid-base reaction with tert-butoxide which I'm just going to draw like this. The tert-butoxide is going to look at that hydrogen and it's going to extract it because it's a strong base. So we're going to take away that hydrogen. But if we make a bond, we have to break a bond. Now this is the interesting part. You haven't really seen many mechanisms that do this. But what we're going to do is we're going to actually place the electrons directly on to the carbon. So essentially instead of going bond to bond, we're going bond to atom which is fine. But now if we make that bond, we have to break another bond. We have to kick out one of the Cls. So we wind up getting as a product of this is now a carbene. What we're going to get is now a carbon with 2 Cls and a lone pair. Does that look familiar? That is my carbene that I was able to create through the elimination reaction. So instead of beta elimination, this would be alpha elimination. But it's very similar concept. Notice the three arrows and we're basically taking away a hydrogen. We're getting rid of 22 single bonds. So now we've got our carbene.
What happens? The mechanism just takes over like before. These arrows are going to be really ugly. I'll erase them in a second. But it would just be this and that. Just the same thing. Now your product is going to be the same exact molecule except what? What do you have to be watchful for? This time we have chlorines, so add those chlorines. Do not forget those chlorines guys. They're important. That's points on your exam. Okay? Awesome. So hopefully that combination made sense.
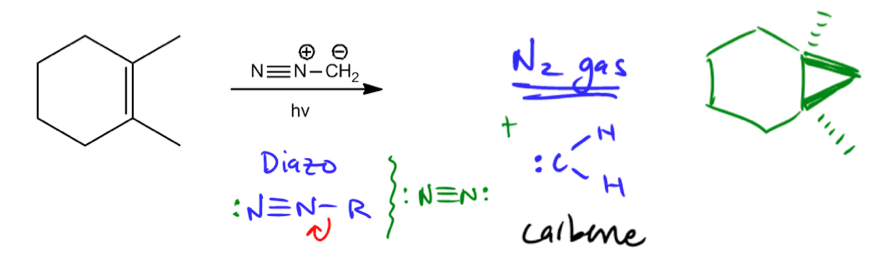
Reaction with diazomethane and light or heat.
Video transcript
Let's move on to the next. So another reagent that creates a cyclopropanation is called diazomethane. Now diazomethane is an interesting structure because first of all, you guys haven't seen this yet. This is more into organic chemistry too. But anytime you see like an NN→ substituent like this, so I'm just going to write it here, N≡N→R, this is called a diazo group. It's just a functional group. It's a function of if we don't teach organic one just because it's kind of beyond the scope of this course. But it's something you will see in the future. And something that's really interesting about diazo groups, guys, is that they love to spontaneously dissociate. Why? Because notice that you've got this N≡N triple bond. And I'm not sure if you guys are aware, nitrogen gas is 78% of the atmosphere is nitrogen gas. It's inert. It's super stable. I mean it's been around for billions of years literally. Nitrogen gas is I'm just going to draw a little squiggly line, is N≡Nlone pair2. This diazo group has a lone pair there. See how close that diazo is to being nitrogen gas? It's almost nitrogen gas. All it has to do is pick up these electrons, take them away and now it takes off into the atmosphere likely to never be reacted again. This thing is going to be like in its nirvana if it can just take those electrons. So guess what allows it to do that? Pretty much any amount of energy. If you insert energy into the system, in this case in terms of light energy, but heat energy could work as well, you'll give the diazo just enough of a little punch to grab those electrons, take off and set sailing for the rest of its happy life. So, this diazomethane when it grabs a little bit of light, guess what's going to ha
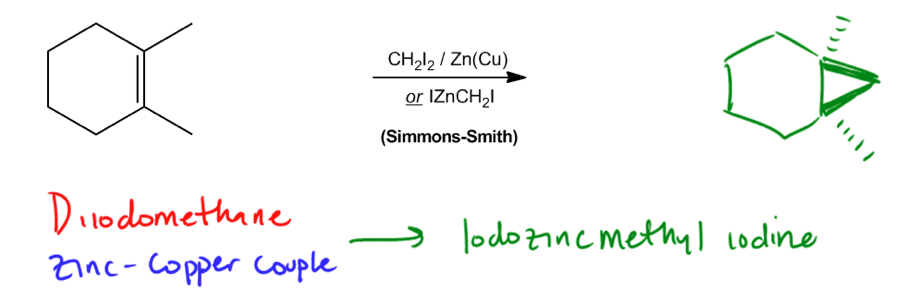
The Simmons-Smith reaction.
Video transcript
Finally, we have the Simmons Smith reaction. Okay? So the Simmons Smith reaction is the most complicated in terms of reagents. I don't need you to memorize all the different combinations as long as you can possibly remember the bottom one. Okay? So the reagents are this. Let me just list them out for you. There's diiodomethane. That's the first one here. So I'm just going to make that one red. There's a zinc copper couple. That's this. And when you react those two things together, guess what happens? When you have the diiodomethane with zinc copper couple, you get them to react together to make something called iodozincmethyl iodide. Literally exactly the way it looks is the way you state the name. And this is what we call the Simmons Smith reagent. The Simmons Smith reagent would be this guy right here. Now that looks really complicated but see the CH2? Guys, that's the important part. The important part is that you're going to make something that looks like this CH
You should not be responsible for the full mechanism of Simmons-Smith, but you should know what the reagents are, and be able to predict that it is a form of cyclopropanation.
Do you want more practice?
More setsYour Organic Chemistry tutors
- When dichloromethane is treated with strong NaOH, an intermediate is generated that reacts like a carbene. D...
- b. Dichlorocarbene can also be generated by heating sodium trichloroacetate. Propose a mechanism for the react...
- Show how you would accomplish each of the following synthetic conversions. b. cyclopentene-->
- Predict the carbenoid addition products of the following reactions. b. + CH2I2, Zn(Cu)
- Predict the carbenoid addition products of the following reactions. a. trans-hex-3-ene + CH2I2, Zn(Cu) b. ci...
- Predict the carbenoid addition products of the following reactions. c. + 50% NaOH/H2O
- Predict the carbenoid addition products of the following reactions. a. cyclohexene + CHCl3, 50% NaOH/H2O
- (••••) In addition to radicals, anions, and cations, a fourth class of reactive intermediates is carbenes. A n...
- [formerly Assessment 8.79(e)] Provide an arrow-pushing mechanism for the cyclopropanation of cyclohexene with ...
- Cyclopropanation using any of the reagents discussed here is stereospecific. (a) What does this say about the...
- Predict the product of the following reactions. (b)
- (••) The following transformation is used as an early step in the synthesis of cilastatin, a drug used to coun...
- Predict the product of the following reactions. (a)
- Cyclopropanation using any of the reagents discussed here is stereospecific. (b) Draw a reaction coordinate d...
- (••••) In addition to radicals, anions, and cations, a fourth class of reactive intermediates is carbenes. A n...
- To determine the stereochemistry of curacin A by synthesis, it would have been necessary to prepare all stereo...
- (•••) Nitrogen-containing heterocycles form particularly stable carbenes and are commonly used as ligands in o...
- The reaction of an alkene with diazomethane forms a cyclopropane ring. Propose a mechanism for the reaction. (...
- Show how you would accomplish each of the following synthetic conversions. a. trans-but-2-ene → trans-1,2-dime...
- Show how you would synthesize each compound using methylenecyclohexane as your starting material. <IMAGE>...
- Propose mechanisms consistent with the following reactions.d. <IMAGE>
- Limonene is one of the compounds that give lemons their tangy odor. Show the structures of the products expect...
- When it is strongly heated, ethyl diazoacetate decomposes to give nitrogen gas and a carbene. Draw a Lewis s...
- Show how you would accomplish each of the following synthetic conversions. c. cyclohexanol-->
- (••••) In addition to radicals, anions, and cations, a fourth class of reactive intermediates is carbenes. A n...
- Suggest a mechanism for the reaction shown in Figure 16.30 using IZnCH₂I as the cyclopropanating reagent.
- Two chemists at Dupont found that ICH2ZnI is better than diazomethane at converting a C'C bond to a cylcopropa...

