Hey everyone, in this video we're going to talk about one of the most important rules in organic chemistry, the octet rule. Now when it comes to atoms, they're the most stable when they achieve the number of electrons necessary to reach what we call the noble gas configuration. All the elements of the periodic table, for the most part, want to obtain a noble gas configuration where they can have their outermost shell completely filled with the optimal number of electrons. If we're to look at a basic periodic table, remember that your noble gases are the elements that are found in group 8A here. So, everything below here would be a noble gas. Now we're going to say that the tendency for atoms to lose or gain electrons in order to reach this configuration is what is known as the octet rule. So most elements, a majority of elements on the periodic table, are trying to reach this noble gas configuration which falls under the idea of the octet rule. Again, this gives the element in question the optimal number of electrons in its outer shell.
Octet Rule - Online Tutor, Practice Problems & Exam Prep
This might be the most important rule in all of undergraduate chemistry! Let’s start from the beginning and go from there.
Noble Gas Configuration
How Noble gases are related to the octet rule.
Video transcript
The tendency for atoms to share electrons to reach their Noble gas configuration is known as the Octet Rule.
LCAO of Noble Gases
LCAO model proves why Noble gases are so stable.
Video transcript
So it turns out that in order to prove that noble gases are so stable, we can actually use the molecular orbital theory that I was teaching you guys before. So let's go ahead. Before I talk about the periodic table more, let's go ahead and go down to this linear combination really quick, and I'm going to show you guys why noble gases are so stable. By the way, think about it this way, if an atom is stable, is it going to want to form a bond or not form a bond? The answer is that it doesn't want to form a bond. The reason is because if it's stable by itself, it doesn't need anyone else. That's like a person that's already financially stable. Right? You have your own, like, amazing apartment. You're not gonna, like, want to get a roommate. Right? You're already, like, thriving. So why would a noble gas get a roommate? And that's exactly the thing. It's not going to. Let me show you why.
So in this case, I'm going to use helium, which if you notice, helium is one of the noble gases, and I'm going to show you guys how it's a little bit different. So remember that these were our atomic orbitals. I'm still using 1sa, 1sb because these are still 1 s orbitals. Remember that one s orbital can hold 2 electrons. How many electrons does each of these helium atoms have? Well, according to the atomic number, what's the atomic number? 2, so it should have 2 electrons. So one sa should have 2 electrons. 1sb should have 2 electrons. Alright? So both of these atomic orbitals are already gonna be full right off the bat. And that's what I've drawn right here. Those are the 2, atoms just nonbonding. If they're nonbonding, they would just interact like that. They wouldn't interact, and they'd just be full already.
Now what happens if we try to make these interact with a bonding or constructive interference? What's going to happen is that 2 of these electrons are going to jump down to a lower energy state. And they're going to fill the sigma molecular orbital as shown below: σ orbital . That's good. The problem is that I still have 2 electrons left that need to go in another molecular orbital because you can't just combine some of them. You have to combine all of them. So then it's going to have to jump up to the next energy state because remember that Pauli exclusion principle says you can only fit 2 in each. And then the principle says that you need to jump up to the next energy level. So what that means is that 2 of these electrons will enter a bonding orbital.
Do you think that's going to be stable? Not at all. That's going to cancel out the stability that was gained from the bonding orbital. So basically, the bonding orbital is going to be canceled out by the full anti-bonding orbital. Does that make sense? So this is really bad.
So it turns out that, I think back in the nineties, they actually did make, finally they got helium to bond. Okay? It took forever. You know how much energy it saved? 0.001 kilojoules per mole. So what that shows you is that really there is basically no benefit to helium interacting through a bonding molecular orbital. So it's not going to. And what you find is that if you do find helium, like in a balloon or in outer space or wherever, helium will be found with just one atom. It won't be found as He2. It will just be found as helium. Alright? So it just shows you how in real life they don't like to bond together. Alright?
The LCAO of He2
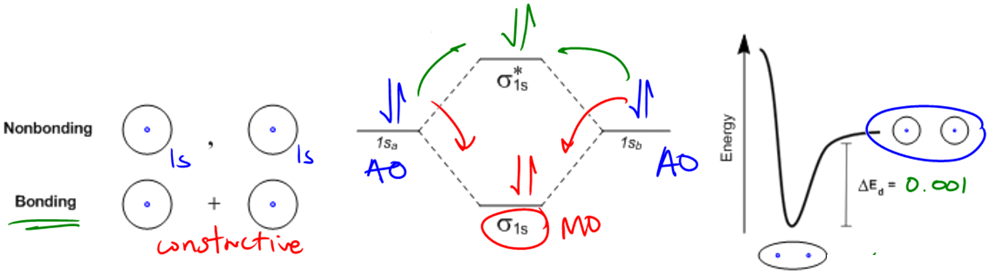
Science-y Version: Helium would have to fill an anti-bonding molecular orbital in order to bond to itself, which undoes the extra stability provided by the filled bonding orbital, so it has no reason to make a bond.
Basic Version: Noble gases don’t like to bond to anything!
Periodic Table Patterns
The most important parts of the periodic table for organic chemistry
Video transcript
So remember we said that organic chemistry is the chemistry of life. So we're not going to see heavy metals involved in organic reactions for the most part or in biological systems. Because of this, the most important rows of the periodic table are rows 12. And sometimes on occasion, you might see row 3 being involved. A vast majority of the reactions will deal with the elements in these three rows. Now, of course, like plants, you wouldn't see a plant composed of atoms of lead, or platinum, or mercury. We will come into contact with reactions, of course, dealing with oxygen and nitrogen and sulfur, things like that.
Now just a crash course in reading this periodic table. We can see that the periodic table has about 118 elements. Now, I haven't filled out everything in terms of this graph, but remember, this bottom row here, it has been completely filled in with conventional names for the elements that are here, here, and here. But again, we're not really concerned with that; we're really focusing on the first three rows.
Now when we talk about rows, remember another name for a row is a period. So, we have row 1, which contains hydrogen and helium, row 2 or period 2, which is lithium, lithium all the way to neon. Row 3 or period 3 is sodium all the way to argon. Besides rows or periods, we have groups. So, our groups represent our columns, or the fancy term families. Now, we'll tend to say groups or columns when we're talking about each one of these groups. So, group 1A here is hydrogen all the way down to Fr, group 2A all the way to group 8A over here.
Now we're not concerning ourselves with the elements found here in the pit. These are your transition metals. They themselves, they're not group A elements, they're transition metals. They're kind of weird. This would actually be, 1B here and 2B here, and then it would crisp it would come back over here as 3B. So transition metals are a bit weird. Just realize that in organic chemistry, we only care mainly about the first three rows or periods, because those are our nonmetal, for the most part, elements. They'll be involved in a lot of reactions you're going to see throughout this course. Okay? So just keep that in mind. Remember, groups are your columns, periods are your rows. We care mainly about the first three rows of the periodic table.
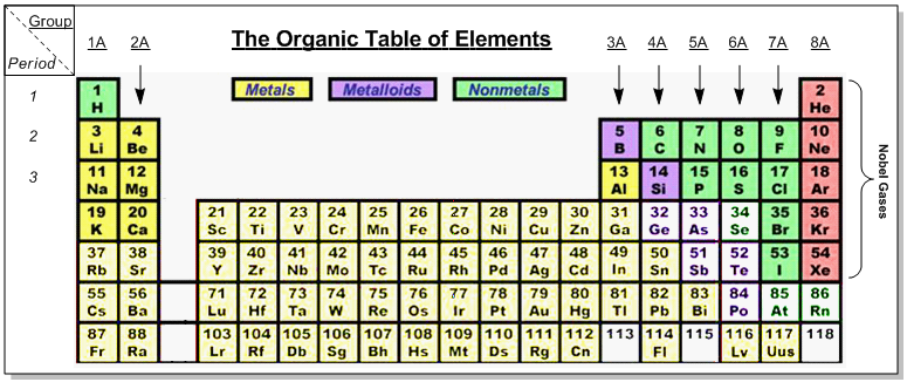
- Only worry about the elements on the first 2 or 3 rows. Living systems don’t have a lot of heavy metals!
- Periods = Rows, Groups = Columns.
- Remember your Group 1A through 8A elements. We will practice memorizing the top atom of each group.
Octet Rule Details
The octet rule.
Video transcript
Now I just want to go over some really quick rules, and these are going to be the rules that we're going to use for the octet rule. Okay? So atoms can satisfy their octet by forming chemical bonds or by possessing lone pairs. So what that basically means is that remember that I said atoms want to either lose electrons or gain electrons in order to fit the noble gas configuration. Right? Well, if you are losing electrons, that means you're going to be sharing electrons with other atoms. Okay? So that would be a bond. If you are gaining electrons, that means you're going to be taking on more electrons so you might have like a lone pair on the atom. Okay? Those electrons are called octet electrons. So keep in mind what an octet electron is. Alright? So basically, let's talk about the first-row elements, hydrogen, helium, and lithium. Okay? Hydrogen, helium, and lithium are so small that they're usually only going to have just that one s orbital and it can only have 2 electrons. Okay? Or they're going to so basically what that means is that they're going to prefer to only have 2 octet electrons so that they can become like helium, okay? So hydrogen is going to want to gain an electron, lithium is going to want to lose an electron, so that they can both be like helium. And they're both going to want basically all of these are going to want to have 2 octet electrons. Cool so far? This is also known in some books as the duet rule. Okay? But the duet rule is the same thing as the octet rule. As long as you think of noble gas, it's the same thing. All right? So then we have our second-row elements. So this is first row or first period you could think of. Second-row elements are carbon, nitrogen, oxygen, and fluorine. These are all going to prefer to possess 8 octet electrons, that's why we use the name octet rule commonly. Now it turns out there is an exception to that. The atoms that are smaller than carbon are going to prefer to possess less than 8 electrons. The reason is because they don't have that many orbitals, so it's actually difficult for them to accommodate 8 electrons. So beryllium is going to prefer to have 4 and boron is going to prefer to have 6. These are just things you need to know. You just need to memorize that. Then finally, we have our third row or third period elements that are going to be able to form what's called expanded octets. Okay? This is what an expanded octet is. We talked about s orbitals and we talked about p orbitals. But it turns out that phosphorus and sulfur are so big that the p orbitals are going to get filled up. Do you know what comes after the p's? The d orbitals. We're not even going to talk about d orbitals in this class. What you should know is that d orbitals can also hold extra electrons. So what's going to happen is that phosphorus is going to be able to hold 10 electrons and sulfur is going to be able to hold 12 electrons.
- First-row elements (H, He, Li) will prefer to possess 2 octet electrons
- Second-row elements (C, N, O, F) will prefer to possess 8 octet electrons
- Atoms smaller than carbon will possess less than 8 electrons:(Be) = 4 and (B) = 6
- Third-row elements may form expanded octets that can hold up to (P) = 10 and (S) = 12 electrons.
Octet electrons is the name we give to ALL electrons that surround an atom. These help the atom reach its Noble gas configuration.
- Bonds = 2 Octet Electrons
- Lone Pairs = 2 Octet Electrons
It seems like a lot to memorize, so let’s just knock out some practice problems.
PRACTICE:Analyze the following molecules. Indicate ALL atoms that are in violation of the octet rule.

From now on let’s just agree that hydrogens with 1 bond and carbons with 4 bonds follow the octet rule, instead of multiplying them out every time.
PRACTICE:Analyze the following molecules. Indicate ALL atoms that are in violation of the octet rule.

You’ll keep seeing the octet rule a bunch more in this chapter. Let’s move on.
Do you want more practice?
More setsYour Organic Chemistry tutors
- How many electrons does an atom of each of the following elements need to lose to achieve a noble gas configur...
- How many electrons does an atom of each of the following elements need to lose to achieve a noble gas configur...
- How many electrons does an atom of each of the following elements need to gain to achieve a noble gas configur...

