Hey everyone, so we're going to start off this course by first talking about the relevance of organic chemistry. At this point, you've taken the course as necessary in general chemistry where you learned about different concepts such as the elements of the periodic table, stoichiometry, titrations, even electrochemistry. The good thing is a majority of this information is not really going to transfer over to organic chemistry. There are only going to be a few concepts mainly around periodic trends that are going to carry over from general chemistry. Right now, don't worry about them too much. We'll cover them when we get to those ideas. Now, back to the relevance of organic chemistry. Well, organic chemistry itself is the chemistry of life. It consists of the study of molecules that are typically created and used by biological systems. So, organic chemistry is important because it involves our everyday lives. If we take a look at this first image, we have the ingredients of a hairspray bottle and I've zeroed in on 4 organic molecules that are found within the ingredients. Right now these molecules don't seem like anything you might recognize. You don't know the number of elements housed within each molecule, but you'll learn about these kinds of ideas and how to interpret a molecule like this later on. But just realize right now that something as simple as a hair bottle is heavily involved in organic chemistry. Now, there are some destructive applications to organic chemistry. Here we have the creation of nerve gases. Here I've circled acetylcholine, acetic acid, and choline itself. These organic molecules are interacting with a biological system here. This is a way of showing how organic chemistry is the chemistry of life, and we're talking about how chemistry interacts with biological systems that also branches off into what we call biochemistry. We're not going to delve too deeply into biochemistry, but just realize that biochemistry is kind of like an extension of these organic chemistry ideas that we're going to discover and talk about this semester. Alright. So for right now just realize that organic chemistry, albeit new to you, is highly relevant in this course and in everyday life.
Intro to Organic Chemistry - Online Tutor, Practice Problems & Exam Prep
Why do we take an entire year learning about Organic chemistry? What makes Organic chemistry so special? Let's find out.
Examples of Organic Molecules
Organic molecules in your everyday life.
Video transcript
Household products have tons of organic molecules in them! It feels awesome when you know what they mean.

Defining Organic Molecules
What is an organic molecule?
Video transcript
So let's just go into recognizing what an organic molecule is. Technically, there are actually a lot of definitions of what an organic molecule is based on the year you're talking about. Back then, they used to have a much different definition. Some books will even say some things that are different. But the majority of the consensus is that an organic molecule, any molecule that contains both carbon and hydrogen, obviously. We know that the chemistry of life is basically built on carbon, at least on Earth, and carbon and hydrogen. The reason I said the whole Earth thing is because some people are like, "Oh, aliens are made out of, like, silicon or something," but, like, whatever. The biology of life has to do with carbons. And then an organic molecule that contains a mixture of hydrogen and carbon only, it doesn't have any other atoms, would be called, you guys know that one? You should know that one. A hydrocarbon.
- Organic Molecule: Any molecule that contains carbon bonded to hydrogen.
- Hydrocarbon: Any molecule that contains only carbon and hydrogen, and nothing else.
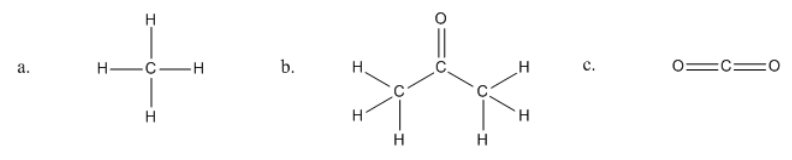
Identifying organic molecules
Video transcript
So hopefully what you said was that letter 'A' is definitely organic. So I'm just going to put an 'O' with a checkbox, and it is because it's made out of carbon and hydrogen. Perfect. Is it also a hydrocarbon? So, I'm going to put the smallest form of carbon called methane. So you might know methane smells bad. It's released like in gas, so sorry. Too much information. But whatever. That is an organic molecule.
How about this next one here? Hopefully, you said that yes, this is organic. Because once again, it has carbon and it has hydrogen. Is it a hydrocarbon? No. This would not be a hydrocarbon. The reason is that it has an oxygen there. See how I have an atom that is not a carbon or a hydrogen? So that means that this is considered just an organic molecule. In fact, this is acetone. So if you, maybe you women use acetone to take your nail polish off, whatever, I hate that smell. It's disgusting. That's what the molecule looks like. We're going to be learning about a lot of really cool molecules. At least, I think they're cool. I have to think they're cool. I'm a tutor. But they are really cool. Hopefully, you'll get to know them too. Don't memorize these names yet. I'm just giving you some information for later.
Then finally, this last one, hopefully, what you said is that this was not organic and also it was not a hydrocarbon. The reason is that I do have a carbon here, but I do not have a hydrogen. There are no hydrogens, so this is not an organic molecule. In fact, this is what we consider can be a little confusing, inorganic carbon, like not organic. This would be a form of inorganic carbon because it doesn't have any hydrogens at all. In fact, this is CO2 or carbon dioxide. So carbon dioxide, you know that's like a greenhouse gas. It goes into the air. That's considered inorganic. We would learn about this a little bit more in gen chem because it has to do more with inorganic and then for organic, we would deal more with the molecules that have hydrogens on them. That's a general rule.
Orgo isn’t so bad, right? Let’s move on to the next topic.
Do you want more practice?
More setsYour Organic Chemistry tutors
- There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part...
- a. Draw resonance contributors for the following species. Do not include structures that are so unstable that ...
- a. Draw resonance contributors for the following species. Do not include structures that are so unstable that ...
- a. Draw resonance contributors for the following species. Do not include structures that are so unstable that ...
- Compound X, isolated from lanolin (sheep’s wool fat), has the pungent aroma of dirty sweatsocks. A careful ana...
- In 1934, Edward A. Doisy of Washington University extracted 3000 lb of hog ovaries to isolate a few milligrams...
- Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propo...
- Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propo...
- Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propo...
- Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propo...

