So now that we understand bonding preferences so well, I want to move to a really related topic called formal charges. And formal charges are just based on the entire idea of bonding preferences. So let's go ahead and just jump right into it. Basically, a formal charge is assigned whenever there's a difference between the number of valence electrons an atom wants to have and the number of valence electrons it actually has. So remember that the group number is how many it wants. The valence electrons, the sticks, and the dots are the amount it actually has. So all you do to calculate formal charge is you take the group number, whatever that is. That could be group 4, group 5, whatever. And then you subtract the valence electrons, which are just the sticks and the dots. It's really simple math. A lot of times you'll just be able to do this on your fingers. Actually, all the time. So you take your group number then you just subtract the sticks and the dots, and you're good. That's called the formal charge. The net charge, let's write that down, the net charge is the term that we give for the sum of all the formal charges. Now this is an important point because I remember when I was in undergrad, I used to get a little confused over like oh, does the formal charge go on the whole thing or is it just one atom? No. The formal charge is 1 atom at a time. You're just looking at each atom and saying, Does this have a formal charge? Does this have a formal charge? The net charge is the collection of all of those summed together. Hopefully, it's not too complicated. There are usually not that many formal charges on a molecule.
Formal Charges - Online Tutor, Practice Problems & Exam Prep
We use our knowledge of valance electrons to determine what he formal charge of the molecule will be.
Formal and Net Charge
Calculating formal and net charge.
Video transcript
Easiest formal charge formula you will find anywhere on the internet:
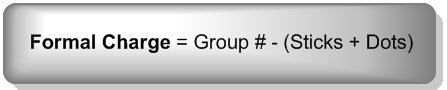
The Net Charge is the sum of all the formal charges on a molecule.
Calculate the formal charge of the following molecule:
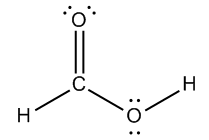
Calculate the formal charges of ALL atoms.
Video transcript
Let's go ahead and do this example where I want to look at each atom and I want to count the formal charges for all of them. What that means is I'm going to be looking at group numbers and I'm going to be looking at sticks and dots. So let's start off with the hydrogens. Even though we already know that this fits the bonding preference. Remember that bonding preferences say that hydrogen wants to have what? One stick. One bond. But let's just do it anyway. Hydrogen is in what group? 1. How many sticks does it have? 1. How many dots does it have? 0. So it's going to be 1 according to my equation. My equation says that formal charge equals group number, which is 1, minus sticks and dots, which is 1. So it's 0. So this has a 0 formal charge. Do you see that? But it also fits with my bonding preference. So as long as your bonding preference agrees with what you see, that's going to be 0.
Let's do this with this oxygen right here. So first of all, does this oxygen fit my bonding preference according to bonding preferences? Yes, it does. It already fits it, so I could already put a 0 here. But now I want to show you mathematically how it works out too. So we said that the formal charge equals the group number. What's the group number of oxygen? It's 6 minus the number of sticks and dots. So how many is that altogether? That's 6. So then I would get a formal charge of 0. See how easy that is?
So now we would do that for carbon, but is carbon fulfilling its bonding preference? It already is. So I can just put a 0 there. Then finally, there was a typo, just so you know. So notice that in your page, the oxygen did not have those lone pairs. Go ahead and just write those in. That just didn't copy. So basically, for this one, the oxygen has 6 electrons total. It wants 6 electrons. I'm sorry. And it has 6 electrons total. In terms of the valence, six sticks and dots, so this would also be 0. This is another way that you can fulfill your bonding preference by having a double bond and having 2 lone pairs. It still counts as 2 bonds and 2 lone pairs. So I hope that you guys see how if it fits its bonding preference, you don't need to calculate. But calculating is always just safe to do.
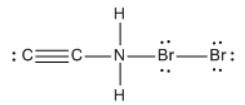
Calculate the net charge of the molecule

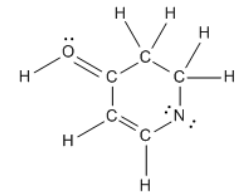
Calculate the total charge of the molecule

Hell yeah! That’s literally all there is to know. Don’t let any professors complicate it more for you.
Do you want more practice?
More setsYour Organic Chemistry tutors
- An atom with a formal charge does not necessarily have more or less electron density than the atoms in the mol...
- An atom with a formal charge does not necessarily have more or less electron density than the atoms in the mol...
- LOOKING AHEAD In Chapter 3, we describe how the formal charge on an atom can be used to predict the number of ...
- (••) By moving an electron pair, draw a better Lewis structure that minimizes formal charges. (b)
- (••) By moving an electron pair, draw a better Lewis structure that minimizes formal charges. (a)
- Draw the Lewis structure for the following ions. Be sure to calculate the formal charge of each atom to confir...
- Calculate the formal charge of the indicated atom in the following molecules or ions. (f)
- Calculate the formal charge of the indicated atom in the following molecules or ions. (e)
- Draw the Lewis structure for the following molecules. Be sure to calculate the formal charge of each atom as a...
- Draw the Lewis structure for the following molecules. Be sure to calculate the formal charge of each atom as a...
- Calculate the formal charge of the indicated atom in the following molecules or ions. (d)
- Draw the Lewis structure for the following ions. Be sure to calculate the formal charge of each atom to confir...
- Draw the Lewis structure for the following ions. Be sure to calculate the formal charge of each atom to confir...
- (a) Calculate the formal charge of the indicated atoms in the ions shown.
- Calculate the formal charge on the non-hydrogen atoms in the molecules shown. Use the arrow-pushing formalism ...
- LOOKING AHEAD In Chapter 3, we describe how the formal charge on an atom can be used to predict the number of ...
- Based on the formal charge, determine how many lone pairs are on each indicated atom. (a)
- Give each atom the appropriate formal charge:a. <IMAGE>b. <IMAGE>
- Draw the missing lone-pair electrons and assign the missing formal charges for the following:c. <IMAGE>d...
- Give each atom the appropriate formal charge:c. <IMAGE>d. <IMAGE>
- Draw the missing lone-pair electrons and assign the missing formal charges for the following:a. <IMAGE>b...
- For each of the following compounds and ions, 1. Draw a Lewis structure. 2. Show the kinds of orbitals that ov...
- For each of the following structures, Draw a Lewis structure; fill in any nonbonding electrons. Calculate the ...
- Draw the Lewis structure for each of the following: d. +C2H5
- A. Draw a Lewis structure for each of the following: 1. CH3N2+ B. Draw a structure for each of the species t...
- Draw the Lewis structure for each of the following: e. CH3NH3+
- (••) Draw the Lewis structure for the following molecular formulas.(u) NH⁺₄
- Draw a Lewis structure for each of the following:b. CO32−

