The following reaction shows the conversion of a diol to a ketone using sulfuric acid:
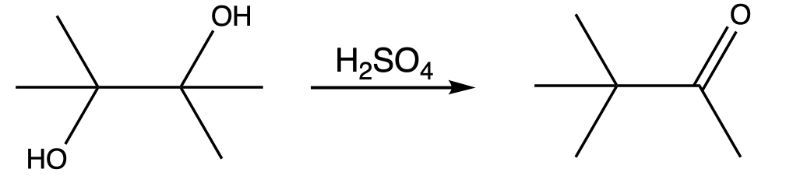
Was the reaction successful based on the IR spectrum of the product? If not, what compound was formed?

Was the reaction successful based on the IR spectrum of the product? If not, what compound was formed?