- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

The gauche conformation of ethane-1,2-diamine (H2N-CH2CH2-NH2) is more stable than its anti-conformation. Draw the Newman projections of these conformers and explain why the gauche conformation is more stable.
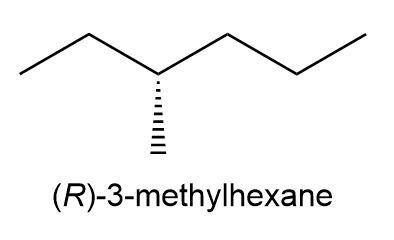
Use an appropriate Newman projection for (R)-3-methylhexane along the C3-C4 bond to draw the most stable conformer.
Provide the Newman projection that would be obtained by looking down the indicated bond in the given direction for the provided structure. Imagine yourself positioned as the eyeball, looking at the bond.

Based on the provided Newman projection, depict the molecule in its line-angle drawing while including all hydrogens and substituents. Ensure that the use of wedges and dashes indicates whether a substituent is projecting out of or receding into the plane of the page. In this structure, note that carbon b is positioned behind carbon a.

Complete the Newman projection based on the initial Newman projection provided and the direction and degree of rotation specified.

Draw the Newman projection for the chair form of cis-1,3-dimethylcyclohexane around the bond between carbon 1 and carbon 6. Keeping both methyl groups in equatorial positions, show that these methyl groups are anti to carbon 5.

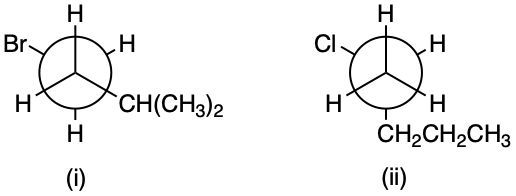
Consider the following Newman projections:
What is their skeletal structure and IUPAC name?