- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

What reagents are required to carry out the reaction shown below?

Draw the product formed when the alkyne below is treated with H2, Pd/C.

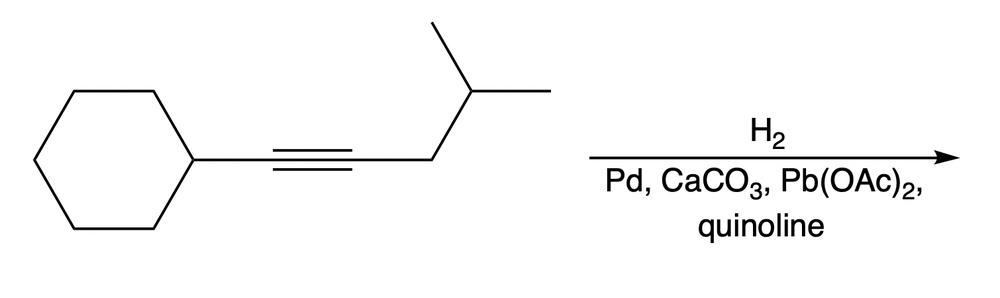
What product would be obtained when the following alkyne is subjected to hydrogenation with a poisoned catalyst?
Show how the following transformations can be accomplished.
(i) oct-2-yne to cis-oct-2-ene.
(ii) hex-2-yne to trans-hex-2-ene.
Propose a mechanism for the acid-catalyzed dehydration of alcohol shown below.

Give the major products formed by dehydration of the given alcohols through sulfuric acid.
Three alkenes are produced when the following seven-membered ring alcohol is dehydrated. Propose a mechanism to explain how they are formed.
Alcohols can undergo dehydration when they are mixed with phosphorus oxychloride (POCl3) in pyridine. In the reaction between an alcohol and phosphorus oxychloride, the alcohol displaces a chloride ion from phosphorus to give an alkyl dichlorophosphate ester. The dichlorophosphate ester then reacts with pyridine base to give an E2 elimination.
Suggest a mechanism for the dehydration of cyclopentanol by POCl3 in pyridine.

Why does the following SN2 reaction of a 1° bromide by alkynide fail to give the desired product?

Show the synthesis of non-4-yne by adding the butyl group and the propyl group afterward. Give all the reagents and intermediates.
Using acetylene as the starting material, show how each of the following compounds can be synthesized. In your synthesis, you can use any compounds with four or less carbon atoms.
(i) trans-but-2-ene
(ii) 1,1-dibromobutane
(iii) 2,2-dibromobutane