- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

The dehydrogenation of ethane (CH3−CH3) to ethene (CH2=CH2) has ΔH° = +136 kJ/mol (+32.5 kcal/mol) and ΔS° = +120 J/kelvin-mol (+28.7 cal/kelvin-mol). Calculate the value of ΔG° for the reaction at room temperature. Is the reaction favored or disfavored?
Cyclopentene undergoes the following light-promoted reaction in the presence of a small amount of bromine.
(i) Write the structure of the rate-limiting transition state.
(ii) Determine which intermediate most closely resembles the rate-limiting transition state based on Hammond's postulate.

Illustrate the representation of the transition state for both the forward and reverse reactions

Determine whether the transition state structures for the given coordinate diagrams are greater in similarity to the reactant's structure or the product's structure.

Briefly explain why ring opening takes place in opposite directions in the reactions shown below.

(a) Arrange the following carbocations in decreasing order of stability.
(b) Determine which carbocation would have the highest rate of formation.
(c) Which carbocation is expected to undergo the fastest reaction with the bromide ion (Br–)?

Rank the following reactive intermediates in order of decreasing reactivity.

Identify the carbocations from the given options that are most likely to undergo rearrangement during a reaction.

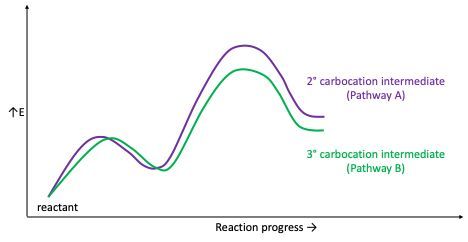
Consider the two pathways for the reaction:
Does the following reaction coordinate for the boxed portion of the two pathways rationalize pathway B as the one that produces the major product?
Show how the following carbocations rearrange to more stable carbocations.