- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

Ethylcyclopentan-1-ylium is more stable than 2-ethylcyclopentan-1-ylium. Provide an energy profile diagram showing that ethylcyclopentan-1-ylium is formed faster than 2-ethylcyclopentan-1-ylium in the following reaction:

Provide an energy profile diagram for an exothermic one-step reaction and label the reactants, products, intermediates, transition states, activation energies (Ea), and ΔG°, if applicable.
It takes several steps to create product I from reactant A. The reaction proceeds as illustrated in the reaction coordinate diagram below.

a. How many intermediates are generated throughout the synthesis?
b. List the letters that correspond to the transition states.
c. Identify which step of the reaction is the fastest.
d. Between A and I, which one is more stable?
e. Does E or I form faster from G?
f. Which intermediate is the most stable?
In the reaction below, which direction would be more favored if temperature increases?

In the acid-base reaction below, determine the ∆G° in kcal/mol at 279 K.

Consider the equilibrium process shown below and its corresponding ∆G°:

(1) What is the equilibrium constant (Keq) of the reaction?
(2) Calculate the % reactants and % products in an equilibrium mixture at 298 K.
Utilizing bond dissociation energies (BDE), determine the change in enthalpy (∆H°) for the reaction given below.

Use the following BDE values:
Determine the value of the standard enthalpy change, ΔH°, for the following reaction:
CH3CH2CH2CH3 + H2 → 2 CH3CH3
BDE values:
CH3CH2—CH2CH3 bond = 88.0 kcal/mol
H—H bond = 104 kcal/mol
CH3CH2—H bond = 101 kcal/mol
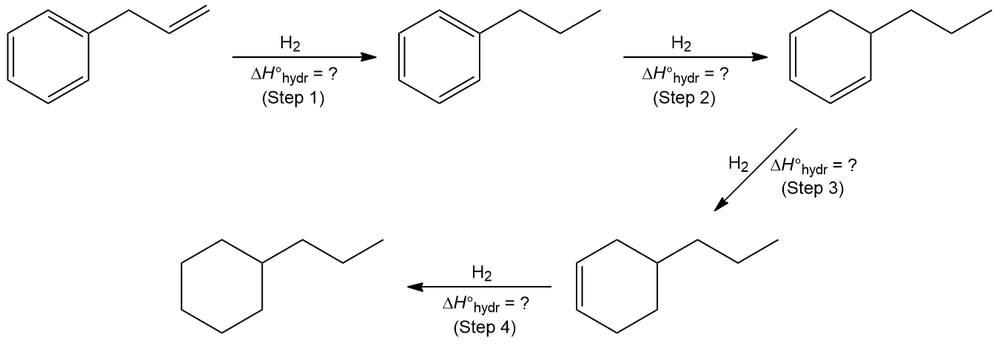
Refer to the information given in the table below. Provide the heat of hydrogenation (∆H°hydr) for each step involved in the reduction of allylbenzene.
∆H°hydr (Step 1) + ∆H°hydr (Step 2) + ∆H°hydr (Step 3) + ∆H°hydr (Step 4) = ∆H°tot = –78.2 kcal/mol (–328 kJ/mol)

Predict the entropy change (ΔS > 0, ΔS = 0, or ΔS < 0) of the following elimination step:

At room temperature the reaction between propene (CH2=CH−CH3) and hydrogen gas (H2) to form propane (CH3−CH2−CH3) goes to completion when a platinum catalyst is present in the reaction mixture.
Determine the signs of ΔH° and ΔS° for this addition reaction based on the information given above.
Explain these signs in terms of freedom of motion and bonding.
CH2=CH−CH3 + H2 (Pt catalyst) → CH3−CH2−CH3