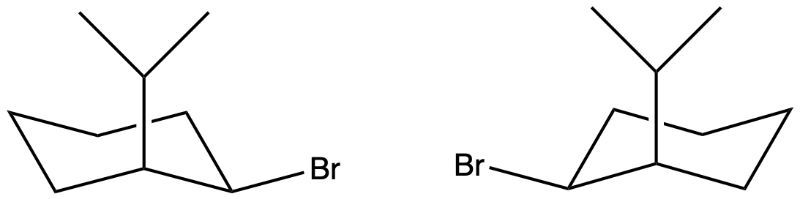- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

Identify if the given pair of molecules are constitutional isomers, stereoisomers, or identical.

Predict if the two molecules below are stereoisomers, constitutional isomers, or identical.
Draw the corresponding three-dimensional structures of (2R,3S)-2,3-dichlorohexane and (1R,2R)-1,2-dichlorocyclohexane.
(i) Write a three-dimensional structure for leucine and 1-chloro-2-methylbutane. Star all asymmetric carbons in the structure you draw.
(ii) Draw the mirror image for each three-dimensional structure, and state whether the mirror images and the structures you drew in part (i) are enantiomers or identical molecules.

Shown in the figure below is the chemical structure of a derivative of Bryostatin 1. (i): How many chiral centers are in this molecule? (ii): Using the number of chiral centers from part (i), calculate the number of possible stereoisomers at these chiral centers. (Ignore stereoisomers at double bonds)

The following racemic mixture of enantiomeric carboxylic acids was synthesized in the laboratory. Propose two methods for the separation of these enantiomers.

60.0 g of compound A was dissolved in 20.0 mL of a solvent and placed in a 1.00-cm polarimeter cell. Calculate the specific rotation of compound A if the observed rotation of the solution is +15.3°.
An unknown compound's (+)-isomer has a specific rotation of +148o. Calculate its observed rotation if a mixture contains 60.0 % (-)-isomer and 40.0 % (+)-isomer.
Consider a theoretical reaction designed to produce one enantiomer by excluding the other. If the difference in activation energies between enantiomer A and enantiomer B is 3.50 kcal/mol and enantiomer B has a lower activation energy, at what temperature would you produce enantiomer B in 99.0 % ee?
Determine whether the following structure is chiral or achiral.

Determine if the given object is achiral or chiral.
Draw the three-dimensional structures of the molecules below. Determine if the compounds have any asymmetric carbons and if either of them are chiral despite not having any asymmetric carbons.
a. (CH3)2C=C=CHBr
b. H2C=C=CHBr
Consider the following compounds:

(i) Give the three-dimensional representation of each compound.
(ii) Does any of the compounds contain a chiral carbon atom?
(iii) Does any of the two compounds not contain a chiral center but is chiral?
Determine whether the following compound is chiral or achiral.

Which of the cyclohexane isomers containing one bromo group and one chloro substituent do not possess any asymmetric centers?