- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

Draw the structure of the 3,5-dimethylphenylhydrazone of propiophenone. Disregard stereoisomers.
What is the product for the reaction of cycloheptanone with phenylhydrazine and weak acid?
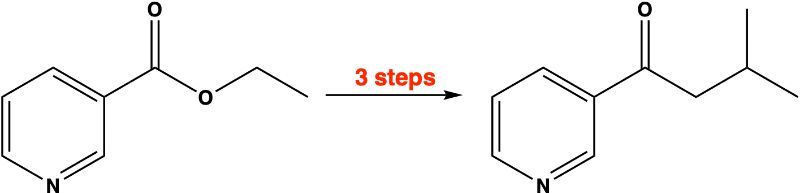
Provide the sequence of reactions that would change the reactant (on the left) into the product (on the right) using a cuprate reagent. It is indicated how many steps are optimum.
Provide the organometallic reagent and nitrile that can be used to produce the given ketone. Note there are two possible ways to make it.

Determine the organometallic reagent and nitrile that can be used to produce the given ketone. Note that there are two possible ways to make it.

Determine the best pair of phosphonium ylide (derived from an alkyl halide) and carbonyl compound for synthesizing the desired compound using the Wittig reaction.

What is the best pair of a phosphonium ylide (derived from an alkyl halide) and a carbonyl compound for synthesizing the given alkene?

Devise a synthesis for the following alkenes using the Wittig reaction starting with an alkyl halide and a ketone or an aldehyde.

Show how hexanal can be synthesized using the starting material given below with any necessary reagent.
- Ethyl hexanoate
Show how 2,3-dimethylhex-2-ene can be converted to pentan-2-one. Provide any necessary reagents.
Show how hexan-1-ol can be converted to hexanal. Provide any necessary reagents.
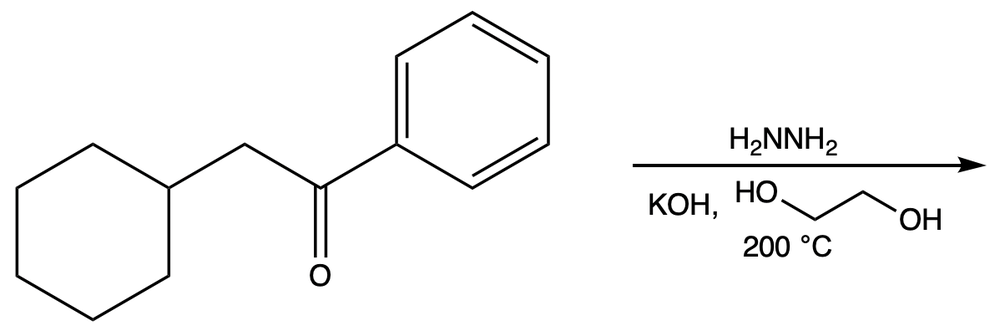
Propose the plausible product(s) for the given reaction. [Note: This reaction is called Wolff–Kishner reduction of aldehydes/ketones.]
Draw the major products expected from the reaction shown below.