- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

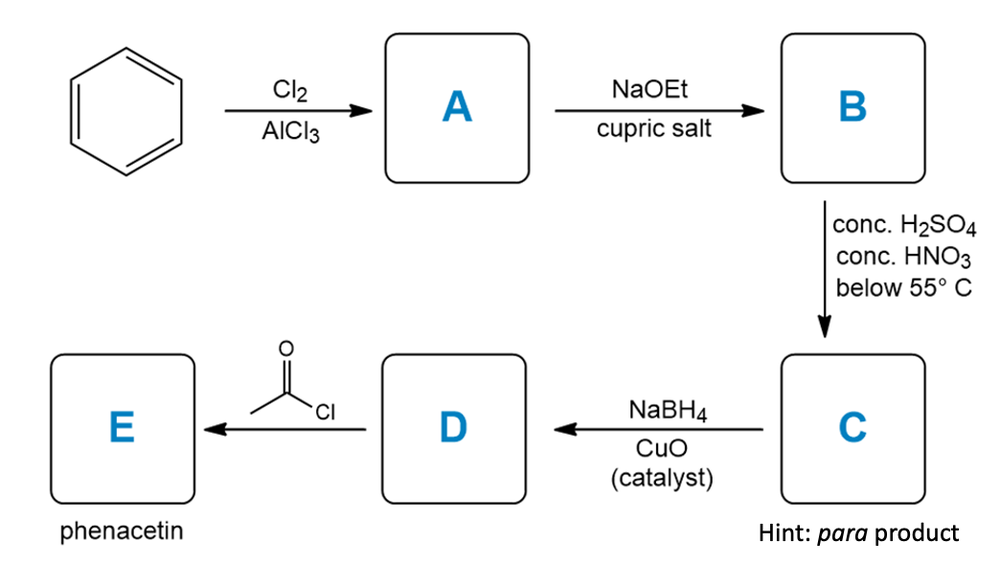
Phenacetin is a medicine used to treat mild to moderate pain. Complete the following synthetic scheme used to synthesize phenacetin:
Note: Hints are provided as needed.
Identify the required reagents for the given reaction sequence.

Provide the correct synthetic sequence for the conversion of ethylbenzene to the given trisubstituted benzene.
Provide the product formed from the reaction shown below.

Draw the structure of the major product expected from the reaction of naphthalen-1-amine with sodium nitrite in dilute hydrochloric acid.
Draw the structure of the diazonium ion and the activated benzene ring used in the synthesis of the following compound.

Identify the reaction mechanisms involved in the following reactions.

Draw the mechanism for the reactions given below.
a. 2,4-dinitrochlorobenzene + ammonia
b. p-nitrobromobenzene + hydrazine (excess)
Consider the following nucleophilic aromatic substitution reaction:

What is the structure of the product formed?
A diazonium group on a benzene ring can be utilized to add an approaching substituent to the meta position. Is this statement true or false? Justify your answer.
Regioselectivity is not usually observed in benzyne mechanisms. However, why does the reaction shown produce a highly regioselective product?

When 1-chloro-2-methylbenzene is treated with sodium amide in ammonia, it undergoes nucleophilic aromatic substitution to form a mixture of two products. Write the products and the mechanism that accounts for the formation of two products.