- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

Arrange the following compounds in ascending order of electrophilic aromatic substitution reactivity.

Draw the resonance contributors of phenoxide and phenol and explain why phenoxide is more reactive than phenol toward EAS.
What is the major product when the compound shown below reacts with Cl2 and AlCl3?

Write a synthetic sequence to convert aniline to the following compound:

Use any reagents necessary to carry out the transformation.
Identify the products formed when the following substituted benzenes undergo nitration.

What will be the product(s) when each of the following compounds is mononitrated?
a. 1,3-dimethylbenzene
b. Salicylic acid
c. 1-bromo-3-chlorobenzene
What are the major products of the reaction of benzene with (i) 4-methylbenzoyl chloride, AlCl3, (ii) 4-chloro-1,2-dimethylcyclopentane, AlCl3, and (iii) 1-chloro-1,4,4-trimethylcyclohexane, AlCl3? If no reaction occurs, write NR.
Arrange the following compounds in decreasing order of reactivity in electrophilic aromatic substitution reactions.

1,4-dimethoxybenzene (0.50 mol) and chlorobenzene (0.50 mol) were mixed together and reacted with 0.50 mol of nitronium ions until all of the nitronium ions were consumed. As a result, two products were obtained in amounts of 0.45 mol and 0.05 mol. Draw the major and minor products.
Identify the correct major product formed when 3-nitrobenzaldehyde reacts with fuming sulfuric acid.

Draw the mechanism for the ortho and para bromination of methoxybenzene.
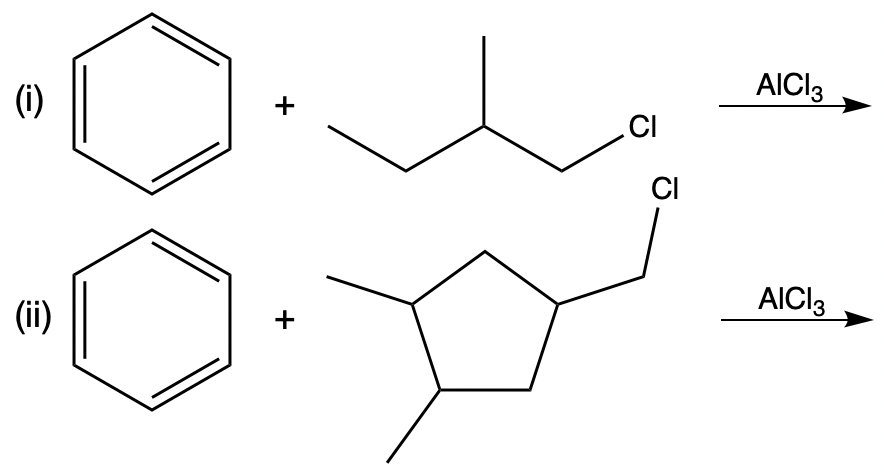
Draw the major products formed if benzene undergoes the Friedel-Crafts alkylation with the alkyl chlorides given below.

Draw the plausible products for the following reactions: