Skip to main content

Determine and explain the mechanism for the cyclopropanation of the given molecule.
The given synthesis is an early step in forming a certain organic molecule. Determine the reagent needed for this step.

Draw the products of the following carbenoid addition reactions.

What is the oxidation state of the indicated atoms in the following structure?

By identifying the oxidation numbers, determine whether the given reaction is a reduction-oxidation reaction.

Determine if each is an oxidation or reduction reaction or if it is neither of the two.
What is the structure of the compound with a molecular formula of C4H8O2 that has the following 1H NMR and IR spectra?
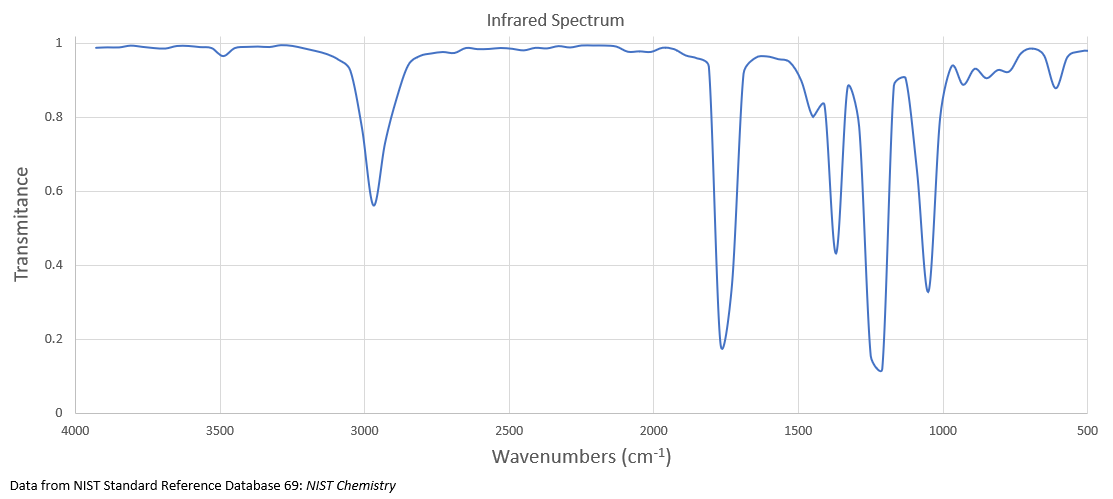

What is the structure of the compound with a molecular formula of C 9H18O that has the following 1H NMR and IR spectra?
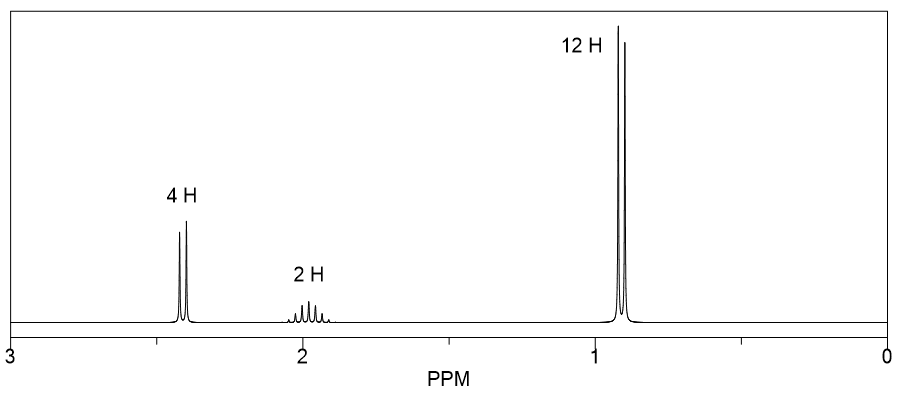

For the following 1H NMR spectrum and the given molecular formula, determine the plausible structure of the organic compound.

(Hint: There is a presence of C=O based on the IR spectrum of this compound that is not shown here.)
Determine the name of the following molecule.

Draw a suitable structure that corresponds to each of the names shown below
(i) α-methylpentanoic acid
(ii) 2-chlorobutanoic acid
(iii) 3-aminopentanoic acid
(iv) cis-5-phenylpent-2-enoic acid