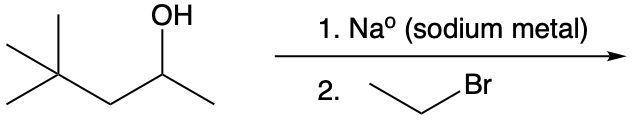- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

Suggest a reaction to convert ethanol into (CH3CH2)2S using a sulfonate ester.
Draw the products that will form when the molecule below reacts with the reagents (i) 1. TsCl, Et3N 2. NaSH, and (ii) 1. TsCl, Et3N 2. NaOt-Bu. Write "no reaction" if no reaction occurs.

Provide the final products of each of the reactions given below.
a. Cyclopentylmethanol + TsCl/pyridine
b. product of (a) +LiAlH4
Identify the ether for which Williamson ether synthesis fails.

Identify the product of the reaction shown below:
Provide a reaction scheme for the syntheses of the following ethers using Williamson ether synthesis. (Note: Use alcohols or phenols as the starting materials.)
(i) 
Suggest a mechanism for the alkoxymercuration reaction below, and box the final product(s).

Propose a plausible mechanism for the oxymercuration-demercuration of this unsaturated alcohol.
Draw a suitable mechanism for the reaction shown below.

A good way to synthesize a symmetrical ether like dipropyl ether is to heat an alcohol with sulfuric acid. Explain why it is not recommended to synthesize an unsymmetrical ether such as butyl isopropyl ether in this manner.
Determine which of the following ethers can be synthesized via condensation of the corresponding alcohols in good yield. Propose a synthesis plan for ethers that can't be made via condensation.
(i) diethyl ether
(ii) methyl ethyl ether
(iii) di-n-propyl ether
One way to synthesize ethers is to use alcohols instead of water as the nucleophiles in the acid-catalyzed hydration. Suggest a mechanism for the conversion of the following alkene into the corresponding ether under the given conditions.