3. Acids and Bases
Acid Base Equilibrium
53PRACTICE PROBLEM
For the given carboxylic acid and protonated amine, determine the pH at which the acid would dissolve in the water layer and the pH at which the protonated amine would dissolve in the ether layer.
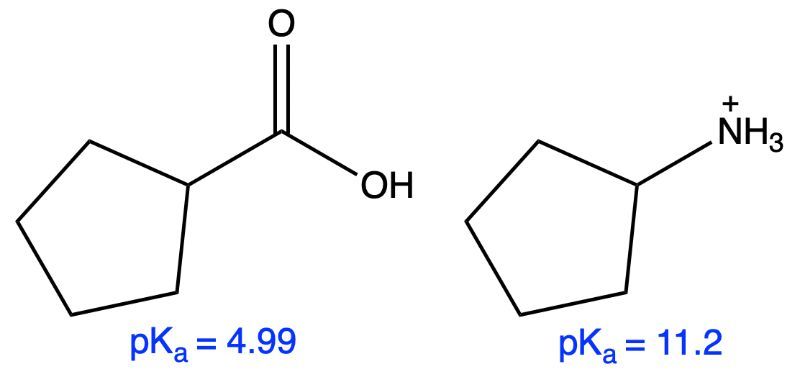
For the given carboxylic acid and protonated amine, determine the pH at which the acid would dissolve in the water layer and the pH at which the protonated amine would dissolve in the ether layer.