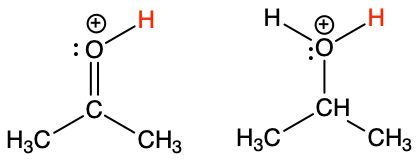
Oxygen shows the same hybridization effect on acidity as carbon and nitrogen.
Each structure below loses an H+ from its positively charged oxygen, resulting in a neutral oxygen. One of the structures has a pKa of −3.2, while the other has a pKa of −7.3.
(i) Show how each reacts with water by writing the chemical reaction.
(ii) Match each structure with its pKa. Justify.