Table of contents
- 1. A Review of General Chemistry(0)
- Summary(0)
- Intro to Organic Chemistry(0)
- Atomic Structure(0)
- Wave Function(0)
- Molecular Orbitals(0)
- Sigma and Pi Bonds(0)
- Octet Rule(0)
- Bonding Preferences(0)
- Formal Charges(0)
- Skeletal Structure(0)
- Lewis Structure(0)
- Condensed Structural Formula(0)
- Degrees of Unsaturation(0)
- Constitutional Isomers(0)
- Resonance Structures(0)
- Hybridization(0)
- Molecular Geometry(0)
- Electronegativity(0)
- 2. Molecular Representations(0)
- 3. Acids and Bases(0)
- 4. Alkanes and Cycloalkanes(0)
- IUPAC Naming(0)
- Alkyl Groups(0)
- Naming Cycloalkanes(0)
- Naming Bicyclic Compounds(0)
- Naming Alkyl Halides(0)
- Naming Alkenes(0)
- Naming Alcohols(0)
- Naming Amines(0)
- Cis vs Trans(0)
- Conformational Isomers(0)
- Newman Projections(0)
- Drawing Newman Projections(0)
- Barrier To Rotation(0)
- Ring Strain(0)
- Axial vs Equatorial(0)
- Cis vs Trans Conformations(0)
- Equatorial Preference(0)
- Chair Flip(0)
- Calculating Energy Difference Between Chair Conformations(0)
- A-Values(0)
- Decalin(0)
- 5. Chirality(0)
- Constitutional Isomers vs. Stereoisomers(0)
- Chirality(0)
- Test 1:Plane of Symmetry(0)
- Test 2:Stereocenter Test(0)
- R and S Configuration(0)
- Enantiomers vs. Diastereomers(0)
- Atropisomers(0)
- Meso Compound(0)
- Test 3:Disubstituted Cycloalkanes(0)
- What is the Relationship Between Isomers?(0)
- Fischer Projection(0)
- R and S of Fischer Projections(0)
- Optical Activity(0)
- Enantiomeric Excess(0)
- Calculations with Enantiomeric Percentages(0)
- Non-Carbon Chiral Centers(0)
- 6. Thermodynamics and Kinetics(0)
- 7. Substitution Reactions(0)
- 8. Elimination Reactions(0)
- 9. Alkenes and Alkynes(0)
- 10. Addition Reactions(0)
- Addition Reaction(0)
- Markovnikov(0)
- Hydrohalogenation(0)
- Acid-Catalyzed Hydration(0)
- Oxymercuration(0)
- Hydroboration(0)
- Hydrogenation(0)
- Halogenation(0)
- Halohydrin(0)
- Carbene(0)
- Epoxidation(0)
- Epoxide Reactions(0)
- Dihydroxylation(0)
- Ozonolysis(0)
- Ozonolysis Full Mechanism(0)
- Oxidative Cleavage(0)
- Alkyne Oxidative Cleavage(0)
- Alkyne Hydrohalogenation(0)
- Alkyne Halogenation(0)
- Alkyne Hydration(0)
- Alkyne Hydroboration(0)
- 11. Radical Reactions(0)
- 12. Alcohols, Ethers, Epoxides and Thiols(0)
- Alcohol Nomenclature(0)
- Naming Ethers(0)
- Naming Epoxides(0)
- Naming Thiols(0)
- Alcohol Synthesis(0)
- Leaving Group Conversions - Using HX(0)
- Leaving Group Conversions - SOCl2 and PBr3(0)
- Leaving Group Conversions - Sulfonyl Chlorides(0)
- Leaving Group Conversions Summary(0)
- Williamson Ether Synthesis(0)
- Making Ethers - Alkoxymercuration(0)
- Making Ethers - Alcohol Condensation(0)
- Making Ethers - Acid-Catalyzed Alkoxylation(0)
- Making Ethers - Cumulative Practice(0)
- Ether Cleavage(0)
- Alcohol Protecting Groups(0)
- t-Butyl Ether Protecting Groups(0)
- Silyl Ether Protecting Groups(0)
- Sharpless Epoxidation(0)
- Thiol Reactions(0)
- Sulfide Oxidation(0)
- 13. Alcohols and Carbonyl Compounds(0)
- 14. Synthetic Techniques(0)
- 15. Analytical Techniques:IR, NMR, Mass Spect(0)
- Purpose of Analytical Techniques(0)
- Infrared Spectroscopy(0)
- Infrared Spectroscopy Table(0)
- IR Spect:Drawing Spectra(0)
- IR Spect:Extra Practice(0)
- NMR Spectroscopy(0)
- 1H NMR:Number of Signals(0)
- 1H NMR:Q-Test(0)
- 1H NMR:E/Z Diastereoisomerism(0)
- H NMR Table(0)
- 1H NMR:Spin-Splitting (N + 1) Rule(0)
- 1H NMR:Spin-Splitting Simple Tree Diagrams(0)
- 1H NMR:Spin-Splitting Complex Tree Diagrams(0)
- 1H NMR:Spin-Splitting Patterns(0)
- NMR Integration(0)
- NMR Practice(0)
- Carbon NMR(0)
- Structure Determination without Mass Spect(0)
- Mass Spectrometry(0)
- Mass Spect:Fragmentation(0)
- Mass Spect:Isotopes(0)
- 16. Conjugated Systems(0)
- Conjugation Chemistry(0)
- Stability of Conjugated Intermediates(0)
- Allylic Halogenation(0)
- Reactions at the Allylic Position(0)
- Conjugated Hydrohalogenation (1,2 vs 1,4 addition)(0)
- Diels-Alder Reaction(0)
- Diels-Alder Forming Bridged Products(0)
- Diels-Alder Retrosynthesis(0)
- Molecular Orbital Theory(0)
- Drawing Atomic Orbitals(0)
- Drawing Molecular Orbitals(0)
- HOMO LUMO(0)
- Orbital Diagram:3-atoms- Allylic Ions(0)
- Orbital Diagram:4-atoms- 1,3-butadiene(0)
- Orbital Diagram:5-atoms- Allylic Ions(0)
- Orbital Diagram:6-atoms- 1,3,5-hexatriene(0)
- Orbital Diagram:Excited States(0)
- Pericyclic Reaction(0)
- Thermal Cycloaddition Reactions(0)
- Photochemical Cycloaddition Reactions(0)
- Thermal Electrocyclic Reactions(0)
- Photochemical Electrocyclic Reactions(0)
- Cumulative Electrocyclic Problems(0)
- Sigmatropic Rearrangement(0)
- Cope Rearrangement(0)
- Claisen Rearrangement(0)
- 17. Ultraviolet Spectroscopy(0)
- 18. Aromaticity(0)
- 19. Reactions of Aromatics: EAS and Beyond(0)
- Electrophilic Aromatic Substitution(0)
- Benzene Reactions(0)
- EAS:Halogenation Mechanism(0)
- EAS:Nitration Mechanism(0)
- EAS:Friedel-Crafts Alkylation Mechanism(0)
- EAS:Friedel-Crafts Acylation Mechanism(0)
- EAS:Any Carbocation Mechanism(0)
- Electron Withdrawing Groups(0)
- EAS:Ortho vs. Para Positions(0)
- Acylation of Aniline(0)
- Limitations of Friedel-Crafts Alkyation(0)
- Advantages of Friedel-Crafts Acylation(0)
- Blocking Groups - Sulfonic Acid(0)
- EAS:Synergistic and Competitive Groups(0)
- Side-Chain Halogenation(0)
- Side-Chain Oxidation(0)
- Reactions at Benzylic Positions(0)
- Birch Reduction(0)
- EAS:Sequence Groups(0)
- EAS:Retrosynthesis(0)
- Diazo Replacement Reactions(0)
- Diazo Sequence Groups(0)
- Diazo Retrosynthesis(0)
- Nucleophilic Aromatic Substitution(0)
- Benzyne(0)
- 20. Phenols(0)
- 21. Aldehydes and Ketones: Nucleophilic Addition(0)
- Naming Aldehydes(0)
- Naming Ketones(0)
- Oxidizing and Reducing Agents(0)
- Oxidation of Alcohols(0)
- Ozonolysis(0)
- DIBAL(0)
- Alkyne Hydration(0)
- Nucleophilic Addition(0)
- Cyanohydrin(0)
- Organometallics on Ketones(0)
- Overview of Nucleophilic Addition of Solvents(0)
- Hydrates(0)
- Hemiacetal(0)
- Acetal(0)
- Acetal Protecting Group(0)
- Thioacetal(0)
- Imine vs Enamine(0)
- Addition of Amine Derivatives(0)
- Wolff Kishner Reduction(0)
- Baeyer-Villiger Oxidation(0)
- Acid Chloride to Ketone(0)
- Nitrile to Ketone(0)
- Wittig Reaction(0)
- Ketone and Aldehyde Synthesis Reactions(0)
- 22. Carboxylic Acid Derivatives: NAS(0)
- Carboxylic Acid Derivatives(0)
- Naming Carboxylic Acids(0)
- Diacid Nomenclature(0)
- Naming Esters(0)
- Naming Nitriles(0)
- Acid Chloride Nomenclature(0)
- Naming Anhydrides(0)
- Naming Amides(0)
- Nucleophilic Acyl Substitution(0)
- Carboxylic Acid to Acid Chloride(0)
- Fischer Esterification(0)
- Acid-Catalyzed Ester Hydrolysis(0)
- Saponification(0)
- Transesterification(0)
- Lactones, Lactams and Cyclization Reactions(0)
- Carboxylation(0)
- Decarboxylation Mechanism(0)
- Review of Nitriles(0)
- 23. The Chemistry of Thioesters, Phophate Ester and Phosphate Anhydrides(0)
- 24. Enolate Chemistry: Reactions at the Alpha-Carbon(0)
- Tautomerization(0)
- Tautomers of Dicarbonyl Compounds(0)
- Enolate(0)
- Acid-Catalyzed Alpha-Halogentation(0)
- Base-Catalyzed Alpha-Halogentation(0)
- Haloform Reaction(0)
- Hell-Volhard-Zelinski Reaction(0)
- Overview of Alpha-Alkylations and Acylations(0)
- Enolate Alkylation and Acylation(0)
- Enamine Alkylation and Acylation(0)
- Beta-Dicarbonyl Synthesis Pathway(0)
- Acetoacetic Ester Synthesis(0)
- Malonic Ester Synthesis(0)
- 25. Condensation Chemistry(0)
- 26. Amines(0)
- 27. Heterocycles(0)
- Nomenclature of Heterocycles(0)
- Acid-Base Properties of Nitrogen Heterocycles(0)
- Reactions of Pyrrole, Furan, and Thiophene(0)
- Directing Effects in Substituted Pyrroles, Furans, and Thiophenes(0)
- Addition Reactions of Furan(0)
- EAS Reactions of Pyridine(0)
- SNAr Reactions of Pyridine(0)
- Side-Chain Reactions of Substituted Pyridines(0)
- 28. Carbohydrates(0)
- Monosaccharide(0)
- Monosaccharides - D and L Isomerism(0)
- Monosaccharides - Drawing Fischer Projections(0)
- Monosaccharides - Common Structures(0)
- Monosaccharides - Forming Cyclic Hemiacetals(0)
- Monosaccharides - Cyclization(0)
- Monosaccharides - Haworth Projections(0)
- Mutarotation(0)
- Epimerization(0)
- Monosaccharides - Aldose-Ketose Rearrangement(0)
- Monosaccharides - Alkylation(0)
- Monosaccharides - Acylation(0)
- Glycoside(0)
- Monosaccharides - N-Glycosides(0)
- Monosaccharides - Reduction (Alditols)(0)
- Monosaccharides - Weak Oxidation (Aldonic Acid)(0)
- Reducing Sugars(0)
- Monosaccharides - Strong Oxidation (Aldaric Acid)(0)
- Monosaccharides - Oxidative Cleavage(0)
- Monosaccharides - Osazones(0)
- Monosaccharides - Kiliani-Fischer(0)
- Monosaccharides - Wohl Degradation(0)
- Monosaccharides - Ruff Degradation(0)
- Disaccharide(0)
- Polysaccharide(0)
- 29. Amino Acids(0)
- Proteins and Amino Acids(0)
- L and D Amino Acids(0)
- Polar Amino Acids(0)
- Amino Acid Chart(0)
- Acid-Base Properties of Amino Acids(0)
- Isoelectric Point(0)
- Amino Acid Synthesis: HVZ Method(0)
- Synthesis of Amino Acids: Acetamidomalonic Ester Synthesis(0)
- Synthesis of Amino Acids: N-Phthalimidomalonic Ester Synthesis(0)
- Synthesis of Amino Acids: Strecker Synthesis(0)
- Reactions of Amino Acids: Esterification(0)
- Reactions of Amino Acids: Acylation(0)
- Reactions of Amino Acids: Hydrogenolysis(0)
- Reactions of Amino Acids: Ninhydrin Test(0)
- 30. Peptides and Proteins(0)
- Peptides(0)
- Primary Protein Structure(0)
- Secondary Protein Structure(0)
- Tertiary Protein Structure(0)
- Disulfide Bonds(0)
- Quaternary Protein Structure(0)
- Summary of Protein Structure(0)
- Intro to Peptide Sequencing(0)
- Peptide Sequencing: Partial Hydrolysis(0)
- Peptide Sequencing: Partial Hydrolysis with Cyanogen Bromide(0)
- Peptide Sequencing: Edman Degradation(0)
- Merrifield Solid-Phase Peptide Synthesis(0)
- 31. Catalysis in Organic Reactions(0)
- 32. Lipids (0)
- 33. The Organic Chemistry of Metabolic Pathways(0)
- Intro to Metabolism(0)
- ATP and Energy(0)
- Intro to Coenzymes(0)
- Coenzymes in Metabolism(0)
- Energy Production in Biochemical Pathways(0)
- Intro to Glycolysis(0)
- Catabolism of Carbohydrates: Glycolysis(0)
- Glycolysis Summary(0)
- Pyruvate Oxidation (Simplified)(0)
- Anaerobic Respiration(0)
- Catabolism of Fats: Glycerol Metabolism(0)
- Intro to Citric Acid Cycle(0)
- Structures of the Citric Acid Cycle(0)
- The Citric Acid Cycle(0)
- 34. Nucleic Acids(0)
- 35. Transition Metals(0)
- Electron Configuration of Elements(0)
- Coordination Complexes(0)
- Ligands(0)
- Electron Counting(0)
- The 18 and 16 Electron Rule(0)
- Cross-Coupling General Reactions(0)
- Heck Reaction(0)
- Stille Reaction(0)
- Suzuki Reaction(0)
- Sonogashira Coupling Reaction(0)
- Fukuyama Coupling Reaction(0)
- Kumada Coupling Reaction(0)
- Negishi Coupling Reaction(0)
- Buchwald-Hartwig Amination Reaction(0)
- Eglinton Reaction(0)
- Catalytic Allylic Alkylation(0)
- Alkene Metathesis(0)
- 36. Synthetic Polymers(0)
- Introduction to Polymers(0)
- Chain-Growth Polymers(0)
- Radical Polymerization(0)
- Cationic Polymerization(0)
- Anionic Polymerization(0)
- Polymer Stereochemistry(0)
- Ziegler-Natta Polymerization(0)
- Copolymers(0)
- Step-Growth Polymers(0)
- Step-Growth Polymers: Urethane(0)
- Step-Growth Polymers: Polyurethane Mechanism(0)
- Step-Growth Polymers: Epoxy Resin(0)
- Polymers Structure and Properties(0)
1. A Review of General Chemistry
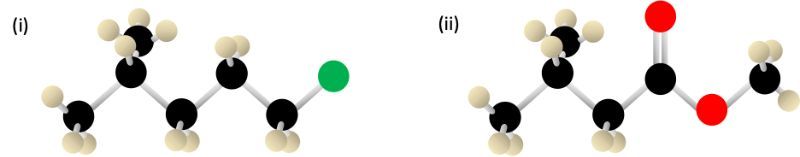
Condensed Structural Formula
1. A Review of General Chemistry
Condensed Structural Formula: Videos & Practice Problems
Condensed Structural Formula Practice Problems
35 problems