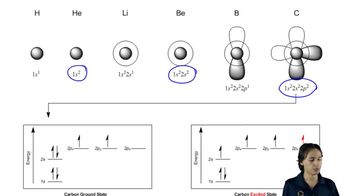
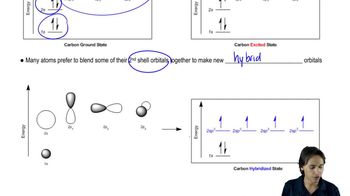
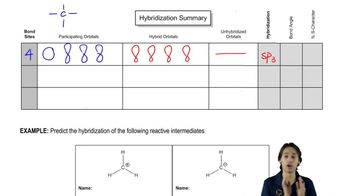
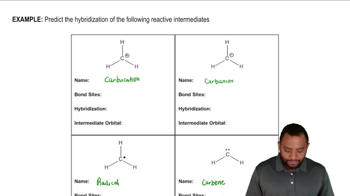
1. A Review of General Chemistry
Hybridization
Problem 1f
Textbook Question
Textbook QuestionExplain why a σ bond formed by overlap of an s orbital with an sp3 orbital of carbon is stronger than a σ bond formed by overlap of an s orbital with a p orbital of carbon.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
576
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos