28. Carbohydrates
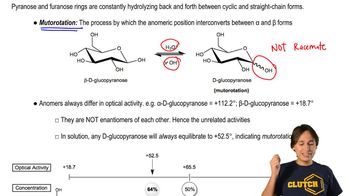
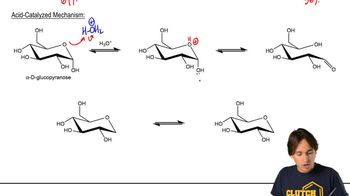
Mutarotation
Problem 20-65
Textbook Question
Textbook QuestionCalculate the percentages of a-d-glucose and b-d-glucose present at equilibrium from the specific rotations of a-d-glucose, b-d-glucose, and the equilibrium mixture. Compare your values with those given in Section 20.10. (Hint: The specific rotation of the mixture equals the specific rotation of a-d-glucose times the fraction of glucose present in the a-form plus the specific rotation of b-d-glucose times the fraction of glucose present in the b-form.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
82
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos