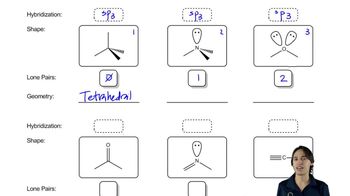
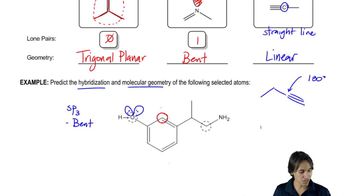
1. A Review of General Chemistry
Molecular Geometry
Problem 1i
Textbook Question
Textbook QuestionIn most amines, the nitrogen atom is sp3 hybridized, with a pyramidal structure and bond angles close to 109°. In urea, both nitrogen atoms are found to be planar, with bond angles close to 120°. Explain this surprising finding. (Hint: Consider resonance forms and the overlap needed in them.) [


 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1287
views
Was this helpful?
Related Videos
Related Practice