24. Enolate Chemistry: Reactions at the Alpha-Carbon
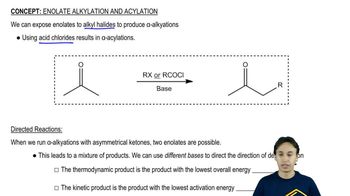
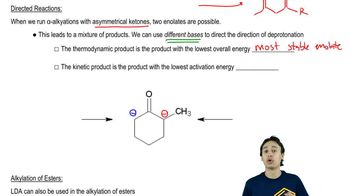
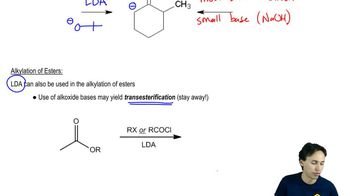
Enolate Alkylation and Acylation
Problem 17a
Textbook Question
Textbook QuestionA carboxylic acid is formed when an a-haloketone reacts with hydroxide ion. This reaction is called a Favorskii reaction. Propose a mechanism for the following Favorskii reaction. (Hint: In the first step, HO- removes a proton from the a-carbon that is not bonded to Br; a three-membered ring is formed in the second step; and HO- is a nucleophile in the third step.)

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
324
views
Was this helpful?
Related Videos
Related Practice