1. A Review of General Chemistry
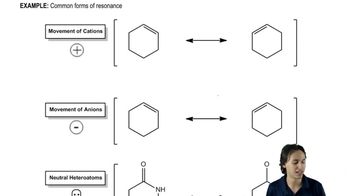
Resonance Structures
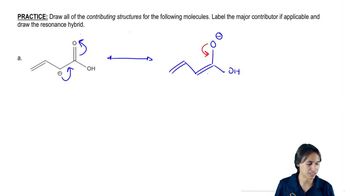
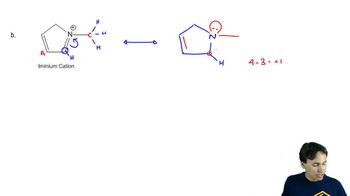
Problem 23.45
Textbook Question
Textbook QuestionAll of the rings of the four heterocyclic bases are aromatic. This is more apparent when the polar resonance forms of the amide groups are drawn, as is done for thymine at here. <IMAGE>
Redraw the hydrogen-bonded guanine-cytosine and adenine-thymine pairs shown in Figure 23-24 <IMAGE>, using the polar resonance forms of the amides. Show how these forms help to explain why the hydrogen bonds involved in these pairings are particularly strong. Remember that a hydrogen bond arises between an electron-deficient hydrogen atom and an electron-rich pair of nonbonding electrons.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
124
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos