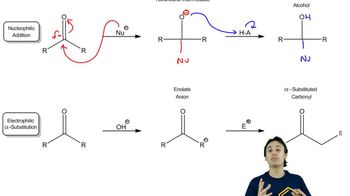
24. Enolate Chemistry: Reactions at the Alpha-Carbon
Enolate
Problem 17c
Textbook Question
Textbook QuestionAlkylation of the following compound with methyl iodide under two different conditions forms two different ketoesters (A and B). Each ketoester forms a cyclic diketone (C and D) when treated with methoxide ion in methanol. a. Draw the structures of A and B, and indicate the conditions used in the alkylation reaction that cause that ketoester to be formed.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
417
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos