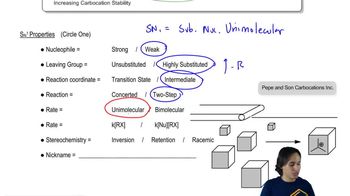
7. Substitution Reactions
SN1 Reaction
Problem 26c
Textbook Question
Textbook QuestionPropose a mechanism involving a hydride shift or an alkyl shift for each solvolysis reaction.
Explain how each rearrangement forms a more stable intermediate.
Hint: Most rearrangements convert 2° (or incipient 1°) carbocations to 3° or resonance-stabilized carbocations.
c. <IMAGE> + CH3C=O(OH) heat—> <IMAGE> + <IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
141
views
Was this helpful?