19. Reactions of Aromatics: EAS and Beyond
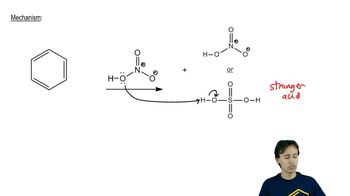
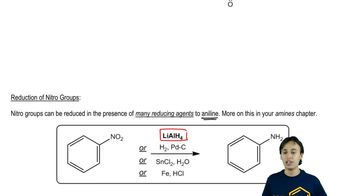
EAS:Nitration Mechanism
Problem 16-37c
Textbook Question
Textbook QuestionBefore spectroscopy was invented, Körner’s absolute method
was used to determine whether a disubstituted
benzene derivative was the ortho, meta, or para isomer.
Körner’s method involves adding a third group
(often a nitro group) and determining how many isomers
are formed. For example, when o-xylene is nitrated
(by a method shown in Chapter 17), two isomers are formed.
<IMAGE of reaction>
(c) A turn-of-the-century chemist isolated
an aromatic compound of molecular formula C6H4Br2.
He carefully nitrated this compound and purified three isomers
of formula C6H3Br2NO2. Propose structures for
the original compound and the three nitrated derivatives.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
76
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos