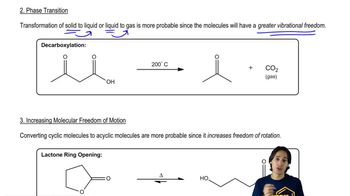
6. Thermodynamics and Kinetics
Entropy
Problem 5-61
Textbook Question
Textbook Questiona. What is the equilibrium constant for a reaction that is carried out at 25 °C (298 K) with ∆H° = 20 kcal/mol and ∆S° = 5.0 * 10 - 2 kcal mol -1K-1?
b. What is the equilibrium constant for the same reaction carried out at 125 °C?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
80
views
Was this helpful?
Related Videos
Related Practice