Textbook Question
What are the major products obtained when each of the following ethers is heated with one equivalent of HI?
e.
1086
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 10 - Reactions of Alcohols, Ethers, Epoxides, Amines, and Sulfur-Containing Compounds
Ch. 10 - Reactions of Alcohols, Ethers, Epoxides, Amines, and Sulfur-Containing Compounds Problem 25
Problem 25 Verified step by step guidance
Verified step by step guidance
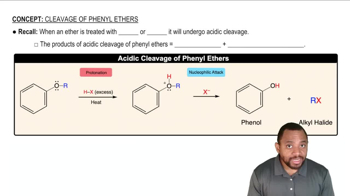


What are the major products obtained when each of the following ethers is heated with one equivalent of HI?
e.
How can the following compounds be prepared from 3,3-dimethyl-1-butene?
b. 3,3-dimethyl-2-butanol
Would you expect the reactivity of a five-membered ring ether such as tetrahydrofuran (Table 10.2) to be more similar to the reactivity of an epoxide or to the reactivity of a noncyclic ether? Why?
What are the major products obtained when each of the following ethers is heated with one equivalent of HI?
c.
d.
What are the major products obtained when the following ether is heated with one equivalent of HI?
Explain why HF and HCl cannot be used to cleave ethers in an SN2 reaction.