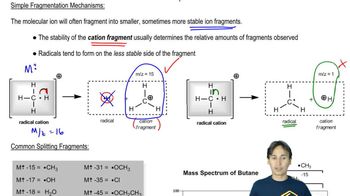
15. Analytical Techniques:IR, NMR, Mass Spect
Mass Spect:Fragmentation
Problem 19a
Textbook Question
Textbook QuestionThe mass spectrum of tert-butylamine follows shows an intense base peak at m/z 58, and very little else. Use a diagram to show the cleavage that accounts for the base peak. Suggest why no molecular ion is visible in this spectrum.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
729
views
Was this helpful?
Related Videos
Related Practice