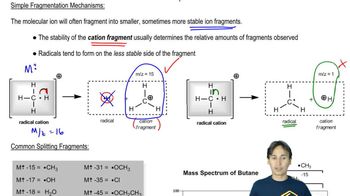
15. Analytical Techniques:IR, NMR, Mass Spect
Mass Spect:Fragmentation
Problem 21
Textbook Question
Textbook QuestionAn unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m/z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is consistent with the observed absorptions. Propose a favorable fragmentation to explain the prominent MS peak at m/z 68.


 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
712
views
Was this helpful?
Related Videos
Related Practice