15. Analytical Techniques:IR, NMR, Mass Spect
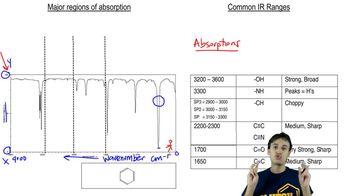
Infrared Spectroscopy
Problem 12a
Textbook Question
Textbook QuestionA C-D (carbon–deuterium) bond is electronically much like a C-H bond, and it has a similar stiffness, measured by the spring constant, k. The deuterium atom has twice the mass (m) of a hydrogen atom, however. (a) The infrared absorption frequency is approximately proportional to the square root of k/m, when one of the bonded atoms is much heavier than the other, and m is the lighter of the two atoms (H or D in this case). Use this relationship to calculate the IR absorption frequency of a typical C-D bond. Use 3000 cm-1 as a typical C-H absorption frequency. (b) A chemist dissolves a sample in deuterochloroform (CDCl3) and then decides to take the IR spectrum and simply evaporates most of the CDCl3. What functional group will appear to be present in this IR spectrum as a result of the CDCl3 impurity?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
14mPlay a video:
993
views
Was this helpful?
Related Videos
Related Practice