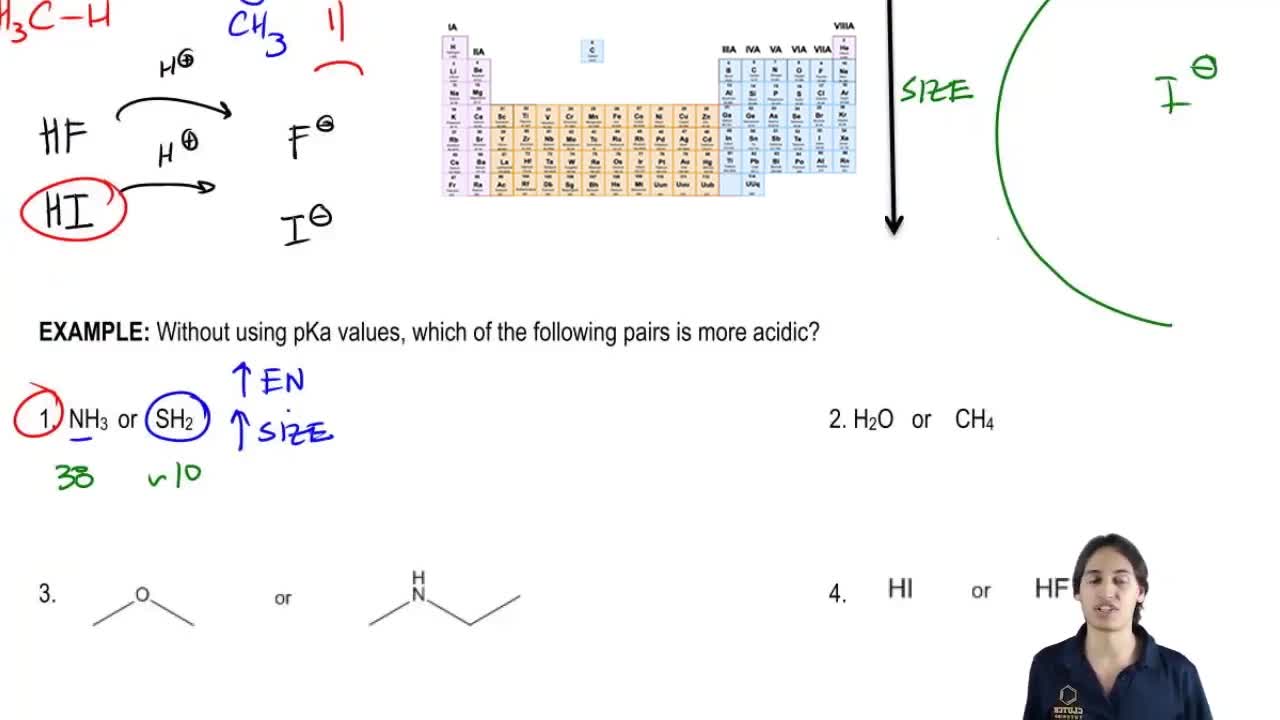
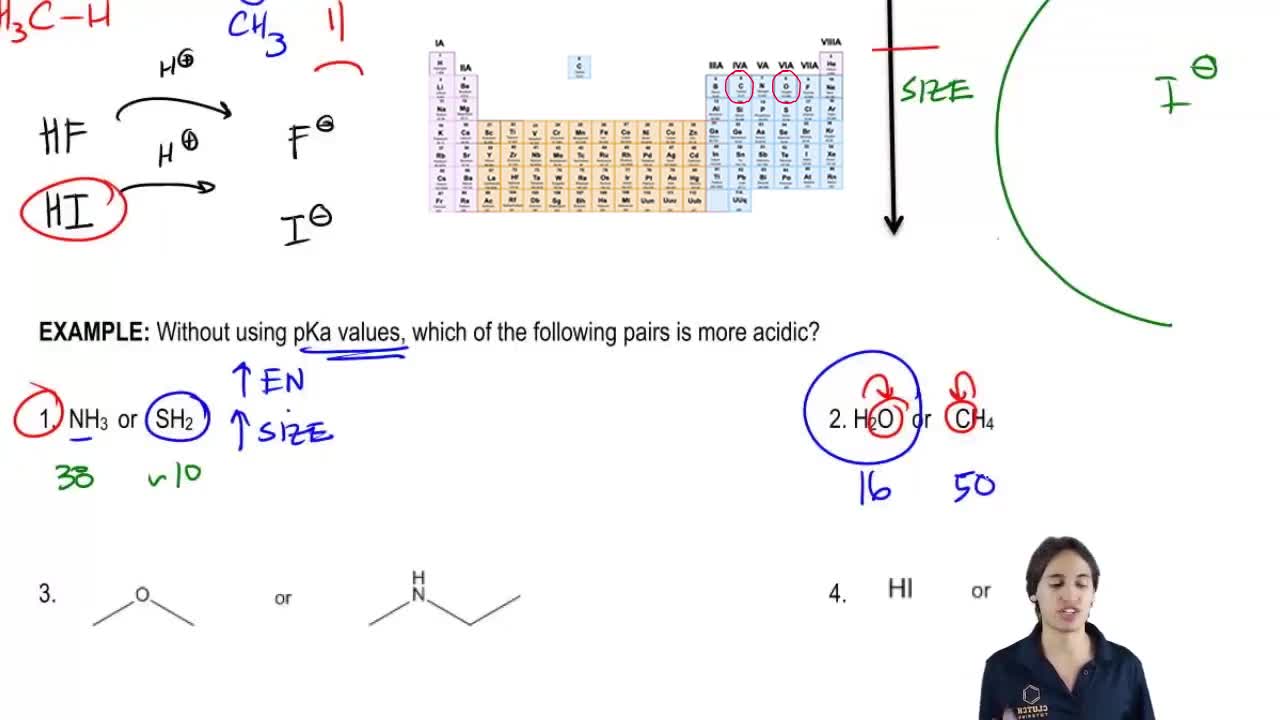
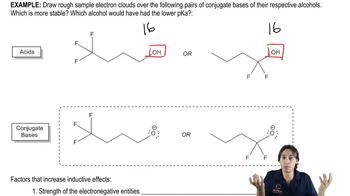
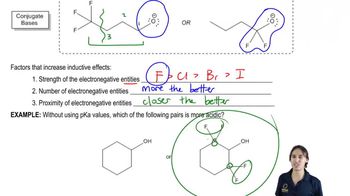
3. Acids and Bases
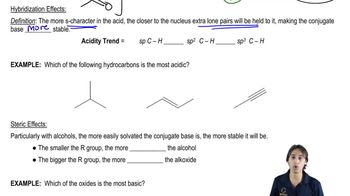
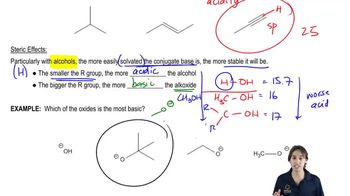
Ranking Acidity
Problem 2j
Textbook Question
Textbook QuestionThe pKa of ascorbic acid (vitamin C, page 55)


 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
2823
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos