Textbook Question
Draw a Lewis Structure for each species.
i. CH3OSO2OCH3
j. CH3C(NH)CH3
k. (CH3)3CNO
1523
views

 Verified step by step guidance
Verified step by step guidance


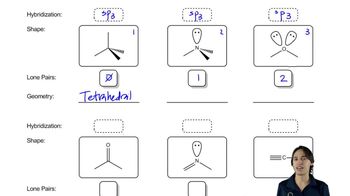
Draw a Lewis Structure for each species.
i. CH3OSO2OCH3
j. CH3C(NH)CH3
k. (CH3)3CNO
Draw a Lewis structure for each compound. Include all nonbonding pairs of electrons.
a. CH3COCH2CHCHCOOH
b. NCCH2COCH2CHO
Draw a line-angle formula for each compound.
c. CH2CHCH(OH)CH2CO2H
d. CH2CHC(CH3)CHCOOCH3
Draw a Lewis Structure for each species.
e. CH3CHO
f. CH3S(O)CH3
g. H2SO4
h. CH3NCO
Draw a complete structural formula and a condensed structural formula for
a. three compounds of formula C3H8O
Draw Lewis structures for
a. two compounds of formula C4H10
b. two compounds of formula C2H6O