3. Acids and Bases
Reaction Mechanism
Problem 2.50i
Textbook Question
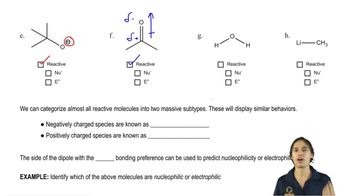
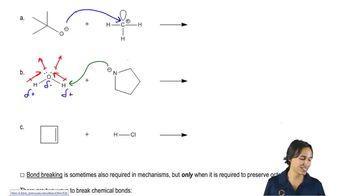
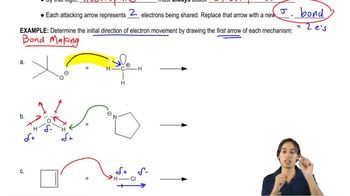
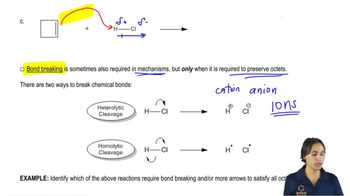
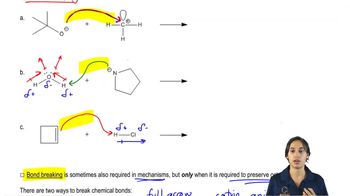
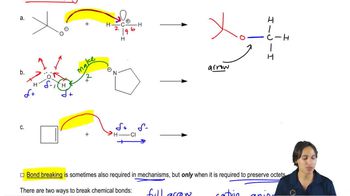
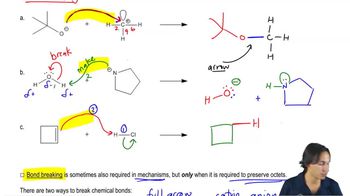
Textbook QuestionLabel the reactants in these acid–base reactions as Lewis acids (electrophiles) or Lewis bases (nucleophiles). Use curved arrows to show the movement of electron pairs in the reactions.
i. -BF3CH2(+CH2) <IMAGE> + CH2CH2 <IMAGE> —> -BF3CH2CH2CH2(+CH2) <IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
124
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos