Textbook Question
Give the important resonance forms for the possible enolate ions of the following:
(d)
668
views

 Wade 9th Edition
Wade 9th Edition Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds
Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds Problem 2b
Problem 2b Verified step by step guidance
Verified step by step guidance
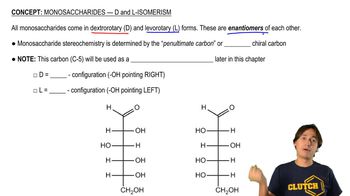
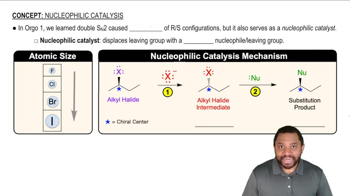
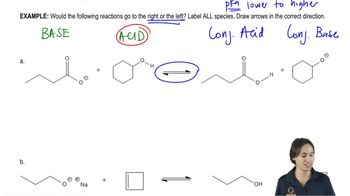
Give the important resonance forms for the possible enolate ions of the following:
(d)
Give the important resonance forms for the possible enolate ions of the following:
(a) acetone
(b) cyclopentanone
Show each step in the mechanism of the acid-catalyzed interconversion of (R)- and (S)-3-methylpentan-2-one.
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the first enol in base.
Give the important resonance forms for the possible enolate ions of the following:
(c) pentane-2,4-dione
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the first enol in acid.