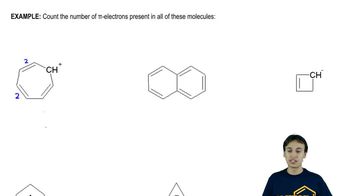
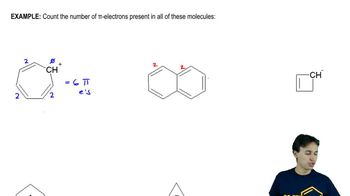
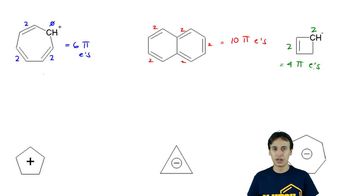
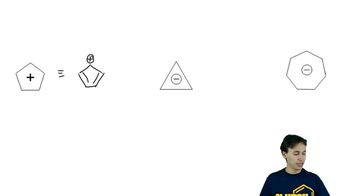
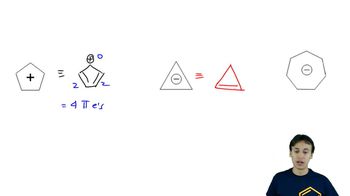18. Aromaticity
Pi Electrons
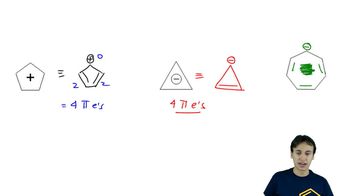
Problem 16-50
Textbook Question
Textbook QuestionChlorophyll is the general name for a family of compounds present in algae and green plants. These molecules use the energy in sunlight to convert carbon dioxide and water into carbohydrates and other energy sources. At the heart of chlorophyll (shown below) is a large-ring magnesium complex called a chlorin. Circle each double bond in the large cyclic conjugated pi system that makes it aromatic. How many pi electrons are in this aromatic system?
<IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
99
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos