6. Thermodynamics and Kinetics
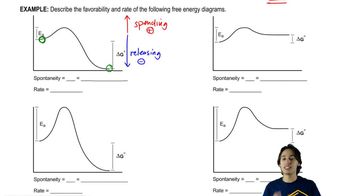
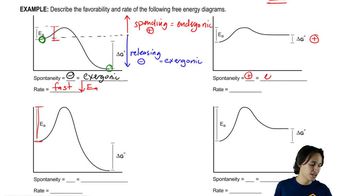
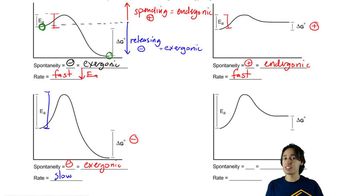
Energy Diagram
Problem 4-39
Textbook Question
Textbook QuestionTreatment of tert-butyl alcohol with concentrated HCl gives tert-butyl chloride.
Tert-butyl alcohol <IMAGE> + H+ + Cl- → tert-butyl chloride <IMAGE> + H2O
When the concentration of H+ is doubled, the reaction rate doubles.
When the concentration of tert-butyl alcohol is tripled, the reaction rate triples.
When the chloride ion concentration is quadrupled, however, the reaction rate is unchanged.
Write the rate equation for this reaction.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
128
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos